Multiple epitopes in the murine cytomegalovirus early gene product M84 are efficiently presented in infected primary macrophages and contribute to strong CD8+-T-lymphocyte responses and protection following DNA immunization
- PMID: 15452242
- PMCID: PMC521820
- DOI: 10.1128/JVI.78.20.11233-11245.2004
Multiple epitopes in the murine cytomegalovirus early gene product M84 are efficiently presented in infected primary macrophages and contribute to strong CD8+-T-lymphocyte responses and protection following DNA immunization
Abstract
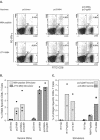
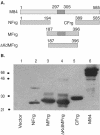
We previously demonstrated that after vaccination of BALB/c mice with DNA encoding murine cytomegalovirus (MCMV) IE1 or M84, a similar level of protection against MCMV infection was achieved. However, the percentage of antigen-specific CD8(+) T cells elicited by IE1 was higher than that by M84 as measured by intracellular cytokine staining when splenocytes were stimulated with an epitope peptide (M. Ye at al., J. Virol. 76:2100-2112, 2002). We show here that after DNA vaccination with M84, a higher percentage of M84-specific CD8(+) T cells was detected when splenocytes were stimulated with J774 cells expressing full-length M84. When the defined M84 epitope 297-305 was deleted, the mutant DNA vaccine was still protective against MCMV replication and induced strong M84-specific CD8(+)-T-cell responses. The M84 gene was subsequently subcloned into three fragments encoding overlapping protein fragments. When mice were immunized with each of the M84 subfragment DNAs, at least two additional protective CD8(+)-T-cell epitopes were detected. In contrast to strong responses after DNA vaccination, M84-specific CD8(+)-T-cell responses were poorly induced during MCMV infection. The weak M84-specific response after MCMV infection was not due to poor antigen presentation in antigen-presenting cells, since both J774 macrophages and primary peritoneal macrophages infected with MCMV in vitro were able to efficiently and constitutively present M84-specific epitopes starting at the early phase of infection. These results indicate that antigen presentation by macrophages is not sufficient for M84-specific CD8(+)-T-cell responses during MCMV infection.
Figures

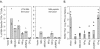


Similar articles
-
Suppression of murine cytomegalovirus (MCMV) replication with a DNA vaccine encoding MCMV M84 (a homolog of human cytomegalovirus pp65).J Virol. 2000 Apr;74(8):3696-708. doi: 10.1128/jvi.74.8.3696-3708.2000. J Virol. 2000. PMID: 10729145 Free PMC article.
-
Strong CD8 T-cell responses following coimmunization with plasmids expressing the dominant pp89 and subdominant M84 antigens of murine cytomegalovirus correlate with long-term protection against subsequent viral challenge.J Virol. 2002 Mar;76(5):2100-12. doi: 10.1128/jvi.76.5.2100-2112.2002. J Virol. 2002. PMID: 11836387 Free PMC article.
-
Development of a vaccine against murine cytomegalovirus (MCMV), consisting of plasmid DNA and formalin-inactivated MCMV, that provides long-term, complete protection against viral replication.J Virol. 2002 May;76(10):4822-35. doi: 10.1128/jvi.76.10.4822-4835.2002. J Virol. 2002. PMID: 11967299 Free PMC article.
-
Intratumoral infection by CMV may change the tumor environment by directly interacting with tumor-associated macrophages to promote cancer immunity.Hum Vaccin Immunother. 2017 Aug 3;13(8):1778-1785. doi: 10.1080/21645515.2017.1331795. Epub 2017 Jun 12. Hum Vaccin Immunother. 2017. PMID: 28604162 Free PMC article. Review.
-
[Induction of apoptosis in murine cytomegalovirus infection].Nihon Rinsho. 1998 Jan;56(1):79-82. Nihon Rinsho. 1998. PMID: 9465669 Review. Japanese.
Cited by
-
DNA immunization using highly conserved murine cytomegalovirus genes encoding homologs of human cytomegalovirus UL54 (DNA polymerase) and UL105 (helicase) elicits strong CD8 T-cell responses and is protective against systemic challenge.J Virol. 2007 Jul;81(14):7766-75. doi: 10.1128/JVI.00633-07. Epub 2007 May 16. J Virol. 2007. PMID: 17507492 Free PMC article.
-
Developing a Vaccine against Congenital Cytomegalovirus (CMV) Infection: What Have We Learned from Animal Models? Where Should We Go Next?Future Virol. 2013 Dec;8(12):1161-1182. doi: 10.2217/fvl.13.106. Future Virol. 2013. PMID: 24523827 Free PMC article.
-
Parameters determining the efficacy of adoptive CD8 T-cell therapy of cytomegalovirus infection.Med Microbiol Immunol. 2012 Nov;201(4):527-39. doi: 10.1007/s00430-012-0258-x. Epub 2012 Sep 13. Med Microbiol Immunol. 2012. PMID: 22972232 Review.
-
CD8 T-cell-based immunotherapy of cytomegalovirus infection: "proof of concept" provided by the murine model.Med Microbiol Immunol. 2008 Jun;197(2):125-34. doi: 10.1007/s00430-008-0093-2. Epub 2008 Mar 15. Med Microbiol Immunol. 2008. PMID: 18343947 Review.
-
Systemic priming-boosting immunization with a trivalent plasmid DNA and inactivated murine cytomegalovirus (MCMV) vaccine provides long-term protection against viral replication following systemic or mucosal MCMV challenge.J Virol. 2005 Jan;79(1):159-75. doi: 10.1128/JVI.79.1.159-175.2005. J Virol. 2005. PMID: 15596812 Free PMC article.
References
-
- Behrens, G., M. Li, C. M. Smith, G. T. Belz, J. Mintern, F. R. Carbone, and W. R. Heath. 2004. Helper T cells, dendritic cells, and CTL immunity. Immunol. Cell. Biol. 82:84-90. - PubMed
-
- Cranmer, L. D., C. L. Clark, C. S. Morello, H. E. Farrell, W. D. Rawlinson, and D. H. Spector. 1996. Identification, analysis, and evolutionary relationship of the putative murine cytomegalovirus homologues of the human cytomegalovirus UL82 (pp71) and UL83 (pp65) matrix phosphoproteins. J. Virol. 70:7929-7939. - PMC - PubMed
-
- Del Val, M., K. Munch, M. J. Reddehase, and U. H. Koszinowski. 1989. Presentation of CMV immediate-early antigen to cytolytic T lymphocytes is selectively prevented by viral genes expressed in the early phase. Cell 58:305-315. - PubMed
-
- Gold, M. C., M. W. Munks, M. Wagner, U. H. Koszinowski, A. B. Hill, and S. P. Fling. 2002. The murine cytomegalovirus immunomodulatory gene m152 prevents recognition of infected cells by M45-specific CTL but does not alter the immunodominance of the M45-specific CD8 T cell response in vivo. J. Immunol. 169:359-365. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

