More than a monolayer: relating lung surfactant structure and mechanics to composition
- PMID: 15454404
- PMCID: PMC1304928
- DOI: 10.1529/biophysj.104.051201
More than a monolayer: relating lung surfactant structure and mechanics to composition
Abstract
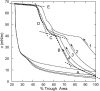
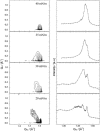
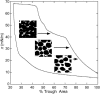
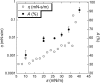
Survanta, a clinically used bovine lung surfactant extract, in contact with surfactant in the subphase, shows a coexistence of discrete monolayer islands of solid phase coexisting with continuous multilayer "reservoirs" of fluid phase adjacent to the air-water interface. Exchange between the monolayer, the multilayer reservoir, and the subphase determines surfactant mechanical properties such as the monolayer collapse pressure and surface viscosity by regulating solid-fluid coexistence. Grazing incidence x-ray diffraction shows that the solid phase domains consist of two-dimensional crystals similar to those formed by mixtures of dipalmitoylphosphatidylcholine and palmitic acid. The condensed domains grow as the surface pressure is increased until they coalesce, trapping protrusions of liquid matrix. At approximately 40 mN/m, a plateau exists in the isotherm at which the solid phase fraction increases from approximately 60 to 90%, at which the surface viscosity diverges. The viscosity is driven by the percolation of the solid phase domains, which depends on the solid phase area fraction of the monolayer. The high viscosity may lead to high monolayer collapse pressures, help prevent atelectasis, and minimize the flow of lung surfactant out of the alveoli due to surface tension gradients.
Figures



Similar articles
-
Keeping lung surfactant where it belongs: protein regulation of two-dimensional viscosity.Biophys J. 2005 Jul;89(1):266-73. doi: 10.1529/biophysj.104.052092. Epub 2005 Apr 15. Biophys J. 2005. PMID: 15833995 Free PMC article.
-
Modifying calf lung surfactant by hexadecanol.Langmuir. 2005 Feb 1;21(3):1028-35. doi: 10.1021/la047894b. Langmuir. 2005. PMID: 15667185
-
Langmuir monolayer of artificial pulmonary surfactant mixtures with an amphiphilic peptide at the air/water interface: comparison of new preparations with surfacten (Surfactant TA).Langmuir. 2008 Apr 1;24(7):3370-9. doi: 10.1021/la703180x. Epub 2008 Mar 4. Langmuir. 2008. PMID: 18315015
-
Characterisation of phase transition in adsorbed monolayers at the air/water interface.Adv Colloid Interface Sci. 2010 Feb 26;154(1-2):1-19. doi: 10.1016/j.cis.2010.01.003. Epub 2010 Jan 28. Adv Colloid Interface Sci. 2010. PMID: 20153454 Review.
-
Role of lipid ordered/disordered phase coexistence in pulmonary surfactant function.Biochim Biophys Acta. 2012 Nov;1818(11):2550-62. doi: 10.1016/j.bbamem.2012.05.024. Epub 2012 May 31. Biochim Biophys Acta. 2012. PMID: 22659676 Review.
Cited by
-
Effect of cholesterol nanodomains on monolayer morphology and dynamics.Proc Natl Acad Sci U S A. 2013 Aug 13;110(33):E3054-60. doi: 10.1073/pnas.1303304110. Epub 2013 Jul 30. Proc Natl Acad Sci U S A. 2013. PMID: 23901107 Free PMC article.
-
Active interfacial shear microrheology of aging protein films.Phys Rev Lett. 2010 Jan 8;104(1):016001. doi: 10.1103/PhysRevLett.104.016001. Epub 2010 Jan 4. Phys Rev Lett. 2010. PMID: 20366371 Free PMC article.
-
Phase transitions of the pulmonary surfactant film at the perfluorocarbon-water interface.Biophys J. 2023 May 16;122(10):1772-1780. doi: 10.1016/j.bpj.2023.04.010. Epub 2023 Apr 11. Biophys J. 2023. PMID: 37041745 Free PMC article.
-
Effect of cholesterol on the molecular structure and transitions in a clinical-grade lung surfactant extract.Proc Natl Acad Sci U S A. 2017 May 2;114(18):E3592-E3601. doi: 10.1073/pnas.1701239114. Epub 2017 Apr 17. Proc Natl Acad Sci U S A. 2017. PMID: 28416656 Free PMC article.
-
Solubility versus electrostatics: what determines lipid/protein interaction in lung surfactant.Biophys J. 2007 Aug 15;93(4):1192-203. doi: 10.1529/biophysj.107.106765. Epub 2007 May 18. Biophys J. 2007. PMID: 17513378 Free PMC article.
References
-
- Alonso, C., and J. Zasadzinski. 2004. Linear dependence of surface drag on surface viscosity. Phys. Rev. E. 69:021602. - PubMed
-
- Als-Nielsen, J., D. Jacquemain, K. Kjaer, L. Lahav, F. Leveiller, and L. Leiserowitz. 1994. Principles and applications of grazing incidence x-ray and neutron scattering from ordered molecular monolayers at the air-water interface. Phys. Rep. 246:251–313.
-
- Als-Nielsen, J., and H. Möhwald. 1991. Synchrotron X-ray Scattering Studies of Langmuir Films. S. Ebashi, M. Koch, and E. Rubinstein, editors. Elsevier, Amsterdam, The Netherlands.
-
- Bangham, A. D., C. J. Morley, and M. C. Phillips. 1979. The physical properties of an effective lung surfactant. Biochim. Biophys. Acta. 573:552–556. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

