Direct comparison of the spread area, contractility, and migration of balb/c 3T3 fibroblasts adhered to fibronectin- and RGD-modified substrata
- PMID: 15454473
- PMCID: PMC1304700
- DOI: 10.1529/biophysj.103.037218
Direct comparison of the spread area, contractility, and migration of balb/c 3T3 fibroblasts adhered to fibronectin- and RGD-modified substrata
Abstract
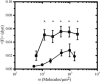
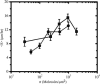
Native proteins are often substituted by short peptide sequences. These peptides can recapitulate key, but not all biofunctional properties of the native proteins. Here, we quantify the similarities and differences in spread area, contractile activity, and migration speed for balb/c 3T3 fibroblasts adhered to fibronectin- (FN) and Arg-Gly-Asp (RGD)-modified substrata of varying surface density. In both cases spread area has a biphasic dependence on surface ligand density (sigma) with a maximum at sigma approximately 200 molecules/microm2, whereas the total traction force increases and reaches a plateau as a function of sigma. In addition to these qualitative similarities, there are significant quantitative differences between fibroblasts adhered to FN and RGD. For example, fibroblasts on FN have a spread area that is on average greater by approximately 200 microm2 over a approximately 40-fold change in sigma. In addition, fibroblasts on FN exert approximately 3-5 times more total force, which reaches a maximum at a value of sigma approximately 5 times less than for cells adhered to RGD. The data also indicate that the differences in traction are not simply a function of the degree of spreading. In fact, fibroblasts on FN (sigma approximately 2000 microm(-2)) and RGD (sigma approximately 200 microm(-2)) have both similar spread area (approximately 600 microm2) and migration speed (approximately 11 microm/h), yet the total force production is five times higher on FN than RGD (approximately 0.05 dyn compared to approximately 0.01 dyn). Thus, the specific interactions between fibroblasts and FN molecules must inherently allow for higher traction force generation in comparison to the interactions between fibroblasts and RGD.
Copyright 2004 Biophysical Society
Figures






References
-
- Akiyama, S. K., E. Hasegawa, T. Hasegawa, and K. M. Yamada. 1985. The interaction of fibronectin fragments with fibroblastic cells. J. Biol. Chem. 260:13256–13260. - PubMed
-
- Aota, S., T. Nagai, and K. M. Yamada. 1991. Characterization of regions of fibronectin besides the arginine-glycine-aspartic acid sequence required for adhesive function of the cell-binding domain using site-directed mutagenesis. J. Biol. Chem. 266:15938–15943. - PubMed
-
- Aota, S., M. Nomizu, and K. M. Yamada. 1994. The short amino acid sequence Pro-His-Ser-Arg-Asn in human fibronectin enhances cell-adhesive function. J. Biol. Chem. 269:24756–24761. - PubMed
-
- Balaban, N. Q., U. S. Schwarz, D. Riveline, P. Goichberg, G. Tzur, I. Sabanay, D. Mahalu, S. Safran, A. Bershadsky, L. Addadi, and B. Geiger. 2001. Force and focal adhesion assembly: a close relationship studied using elastic micropatterned substrates. Nat. Cell. Biol. 3:466–472. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

