Efficacy of two once-daily methylphenidate formulations compared across dose levels at different times of the day: preliminary indications from a secondary analysis of the COMACS study data
- PMID: 15458569
- PMCID: PMC524494
- DOI: 10.1186/1471-244X-4-28
Efficacy of two once-daily methylphenidate formulations compared across dose levels at different times of the day: preliminary indications from a secondary analysis of the COMACS study data
Abstract
Background: Methylphenidate (MPH) is commonly prescribed in the treatment of Attention-Deficit/Hyperactivity Disorder or ADHD. Concerta and Metadate CD are once-daily formulations of MPH using different delivery mechanisms resulting in different pharmacokinetic profiles. A recent study (COMACS) showed that for near-milligram (mg) equivalent daily doses, Metadate CD provides greater symptom control in the morning (1.5 through 4.5 hours post-dose), while Concerta provides greater control in the early evening (12 hours post-dose). Non-inferential comparison of effects for different dose levels of the two formulations suggested that equivalent levels of morning symptom control could be obtained with lower daily doses of Metadate CD than Concerta; the situation being reversed in the evening. The current paper presents a secondary analysis that provides a statistical test of these observations.
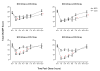
Method: The COMACS study was a multi-center, double-blind crossover study of Metadate CD, Concerta and placebo with each treatment administered for 1 week. Children were assigned on the basis of their pre-trial dosage to either high (Metadate CD 60 mg; Concerta 54 mg), medium (Metadate CD 40 mg; Concerta 36 mg) or low doses (Metadate CD 20 mg; Concerta 18 mg) of MPH, and attended a laboratory school on the 7th day for assessment at 7 sessions across the day. For the post-hoc comparisons across dose levels presented here, total SKAMP scores with the active treatments (adjusted for placebo response) were analyzed using an analysis of covariance, with a combined measure modeling placebo response across all time period as the covariate.
Results: Symptom control from 1.5 through 6.0 hours post-dose was as good with lower doses of Metadate CD (20 and 40 mg) as with higher doses of Concerta (36 and 54 mg, respectively). Lower daily doses of Concerta (18 and 36 mg) and higher doses of Metadate CD (40 and 60 mg, respectively) gave equivalent control at 7.5 and 12 hours with Metadate CD giving better control from1.5 through 6.0 hours post-dose.
Conclusions: Different delivery profiles of Metadate CD and Concerta can be exploited to limit total daily exposure to MPH while at the same targeting a specific, especially clinically significant, period of the day. These results need to be confirmed in a study in which children are randomly allocated to different dose levels of the two formulations and plasma MPH concentrations are assessed simultaneously.
Figures
References
-
- Mannuzza S, Klein RG. Long-term prognosis in attention deficit/hyperactivity disorder. Child Adolesc Psychiatr Clin N Am. 2000;9:711–726. - PubMed
-
- American Academy of Pediatrics, Subcommittee on Attention-Deficit/Hyperactivity Disorder, Committee on Quality Improvement Clinical practice guideline: Treatment of the school-aged child with Attention-Defecit/Hyperactivity Disorder. Pediatrics. 2001;108:1033–1044. doi: 10.1542/peds.108.4.1033. - DOI - PubMed
-
- Horrigan JP, Kohli RR. The impact of dosing frequency on psychostimulant compliance in ADHD. Presented at the 42nd Annual New Clinical Drug Evaluation Unit (NCDEU) meeting: Boca Raton. http://www.nimh.nih.gov/ncdeu/abstracts2002/ncdeu2056.cfm 10–13 June 2002.