A naturally occurring mutation of the opsin gene (T4R) in dogs affects glycosylation and stability of the G protein-coupled receptor
- PMID: 15459196
- PMCID: PMC1351288
- DOI: 10.1074/jbc.M408472200
A naturally occurring mutation of the opsin gene (T4R) in dogs affects glycosylation and stability of the G protein-coupled receptor
Abstract
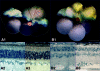
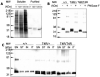
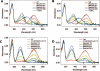
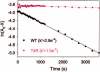
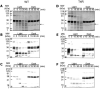
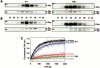
Rho (rhodopsin; opsin plus 11-cis-retinal) is a prototypical G protein-coupled receptor responsible for the capture of a photon in retinal photoreceptor cells. A large number of mutations in the opsin gene associated with autosomal dominant retinitis pigmentosa have been identified. The naturally occurring T4R opsin mutation in the English mastiff dog leads to a progressive retinal degeneration that closely resembles human retinitis pigmentosa caused by the T4K mutation in the opsin gene. Using genetic approaches and biochemical assays, we explored the properties of the T4R mutant protein. Employing immunoaffinity-purified Rho from affected RHO(T4R/T4R) dog retina, we found that the mutation abolished glycosylation at Asn(2), whereas glycosylation at Asn(15) was unaffected, and the mutant opsin localized normally to the rod outer segments. Moreover, we found that T4R Rho(*) lost its chromophore faster as measured by the decay of meta-rhodopsin II and that it was less resistant to heat denaturation. Detergent-solubilized T4R opsin regenerated poorly and interacted abnormally with the G protein transducin (G(t)). Structurally, the mutation affected mainly the "plug" at the intradiscal (extracellular) side of Rho, which is possibly responsible for protecting the chromophore from the access of bulk water. The T4R mutation may represent a novel molecular mechanism of degeneration where the unliganded form of the mutant opsin exerts a detrimental effect by losing its structural integrity.
Figures





Similar articles
-
Assessment of visual function and retinal structure following acute light exposure in the light sensitive T4R rhodopsin mutant dog.Exp Eye Res. 2016 May;146:341-353. doi: 10.1016/j.exer.2016.04.006. Epub 2016 Apr 13. Exp Eye Res. 2016. PMID: 27085210 Free PMC article.
-
P23H opsin knock-in mice reveal a novel step in retinal rod disc morphogenesis.Hum Mol Genet. 2014 Apr 1;23(7):1723-41. doi: 10.1093/hmg/ddt561. Epub 2013 Nov 7. Hum Mol Genet. 2014. PMID: 24214395 Free PMC article.
-
Pharmacological chaperone-mediated in vivo folding and stabilization of the P23H-opsin mutant associated with autosomal dominant retinitis pigmentosa.J Biol Chem. 2003 Apr 18;278(16):14442-14450. doi: 10.1074/jbc.M300087200. Epub 2003 Feb 1. J Biol Chem. 2003. PMID: 12566452 Free PMC article.
-
Structural aspects of rod opsin and their implication in genetic diseases.Pflugers Arch. 2021 Sep;473(9):1339-1359. doi: 10.1007/s00424-021-02546-x. Epub 2021 Mar 16. Pflugers Arch. 2021. PMID: 33728518 Review.
-
Role of membrane integrity on G protein-coupled receptors: Rhodopsin stability and function.Prog Lipid Res. 2011 Jul;50(3):267-77. doi: 10.1016/j.plipres.2011.03.002. Epub 2011 Mar 22. Prog Lipid Res. 2011. PMID: 21435354 Free PMC article. Review.
Cited by
-
Autosomal recessive retinitis pigmentosa E150K opsin mice exhibit photoreceptor disorganization.J Clin Invest. 2013 Jan;123(1):121-37. doi: 10.1172/JCI66176. Epub 2012 Dec 10. J Clin Invest. 2013. PMID: 23221340 Free PMC article.
-
Exclusion of the unfolded protein response in light-induced retinal degeneration in the canine T4R RHO model of autosomal dominant retinitis pigmentosa.PLoS One. 2015 Feb 19;10(2):e0115723. doi: 10.1371/journal.pone.0115723. eCollection 2015. PLoS One. 2015. PMID: 25695253 Free PMC article.
-
Pharmacological and rAAV gene therapy rescue of visual functions in a blind mouse model of Leber congenital amaurosis.PLoS Med. 2005 Nov;2(11):e333. doi: 10.1371/journal.pmed.0020333. Epub 2005 Nov 1. PLoS Med. 2005. PMID: 16250670 Free PMC article.
-
Mutation-independent rhodopsin gene therapy by knockdown and replacement with a single AAV vector.Proc Natl Acad Sci U S A. 2018 Sep 4;115(36):E8547-E8556. doi: 10.1073/pnas.1805055115. Epub 2018 Aug 20. Proc Natl Acad Sci U S A. 2018. PMID: 30127005 Free PMC article.
-
Study of a synthetic human olfactory receptor 17-4: expression and purification from an inducible mammalian cell line.PLoS One. 2008 Aug 6;3(8):e2920. doi: 10.1371/journal.pone.0002920. PLoS One. 2008. PMID: 18682799 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- EY13729/EY/NEI NIH HHS/United States
- EY08061/EY/NEI NIH HHS/United States
- R01 EY006855/EY/NEI NIH HHS/United States
- R01 EY008061/EY/NEI NIH HHS/United States
- EY06855/EY/NEI NIH HHS/United States
- R01 EY013132/EY/NEI NIH HHS/United States
- P30 EY001730/EY/NEI NIH HHS/United States
- EY01730/EY/NEI NIH HHS/United States
- U10 EY013729/EY/NEI NIH HHS/United States
- GM63020/GM/NIGMS NIH HHS/United States
- R01 GM063020/GM/NIGMS NIH HHS/United States
- EY13385/EY/NEI NIH HHS/United States
- EY13132/EY/NEI NIH HHS/United States
- R01 EY013385/EY/NEI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

