The zebrafish nrc mutant reveals a role for the polyphosphoinositide phosphatase synaptojanin 1 in cone photoreceptor ribbon anchoring
- PMID: 15470129
- PMCID: PMC6729946
- DOI: 10.1523/JNEUROSCI.2892-04.2004
The zebrafish nrc mutant reveals a role for the polyphosphoinositide phosphatase synaptojanin 1 in cone photoreceptor ribbon anchoring
Abstract
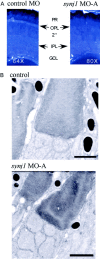
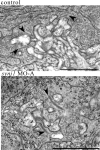
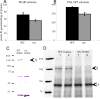
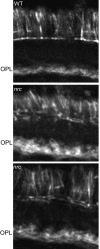
Visual, vestibular, and auditory neurons rely on ribbon synapses for rapid continuous release and recycling of synaptic vesicles. Molecular mechanisms responsible for the properties of ribbon synapses are mostly unknown. The zebrafish vision mutant nrc has unanchored ribbons and abnormal synaptic transmission at cone photoreceptor synapses. We used positional cloning to identify the nrc mutation as a premature stop codon in the synaptojanin1 (synj1) gene. Synaptojanin 1 (Synj1) is undetectable in nrc extracts, and biochemical activities associated with it are reduced. Furthermore, morpholinos directed against synj1 phenocopy the nrc mutation. Synj1 is a polyphosphoinositide phosphatase important at conventional synapses for clathrin-mediated endocytosis and actin cytoskeletal rearrangement. In the nrc cone photoreceptor pedicle, not only are ribbons unanchored, but synaptic vesicles are reduced in number, abnormally distributed, and interspersed within a dense cytoskeletal matrix. Our findings reveal a new role for Synj1 and link phosphoinositide metabolism to ribbon architecture and function at the cone photoreceptor synapse.
Figures



Similar articles
-
Differential role for synaptojanin 1 in rod and cone photoreceptors.J Comp Neurol. 2009 Dec 10;517(5):633-44. doi: 10.1002/cne.22176. J Comp Neurol. 2009. PMID: 19827152 Free PMC article.
-
Synaptojanin1 is required for temporal fidelity of synaptic transmission in hair cells.PLoS Genet. 2009 May;5(5):e1000480. doi: 10.1371/journal.pgen.1000480. Epub 2009 May 8. PLoS Genet. 2009. PMID: 19424431 Free PMC article.
-
Synaptojanin 1 is required for endolysosomal trafficking of synaptic proteins in cone photoreceptor inner segments.PLoS One. 2014 Jan 2;9(1):e84394. doi: 10.1371/journal.pone.0084394. eCollection 2014. PLoS One. 2014. PMID: 24392132 Free PMC article.
-
Ribbon synapses: anchors away for a fishy tale.Curr Biol. 2005 Feb 8;15(3):R102-5. doi: 10.1016/j.cub.2005.01.030. Curr Biol. 2005. PMID: 15694286 Review.
-
Ribbon synapses in zebrafish hair cells.Hear Res. 2015 Dec;330(Pt B):170-7. doi: 10.1016/j.heares.2015.04.003. Epub 2015 Apr 25. Hear Res. 2015. PMID: 25916266 Free PMC article. Review.
Cited by
-
Differential role for synaptojanin 1 in rod and cone photoreceptors.J Comp Neurol. 2009 Dec 10;517(5):633-44. doi: 10.1002/cne.22176. J Comp Neurol. 2009. PMID: 19827152 Free PMC article.
-
Harmonin (Ush1c) is required in zebrafish Müller glial cells for photoreceptor synaptic development and function.Dis Model Mech. 2011 Nov;4(6):786-800. doi: 10.1242/dmm.006429. Epub 2011 Jul 14. Dis Model Mech. 2011. PMID: 21757509 Free PMC article.
-
The dual phosphatase activity of synaptojanin1 is required for both efficient synaptic vesicle endocytosis and reavailability at nerve terminals.Neuron. 2007 Dec 20;56(6):1004-18. doi: 10.1016/j.neuron.2007.10.032. Neuron. 2007. PMID: 18093523 Free PMC article.
-
Rapid synaptic vesicle endocytosis in cone photoreceptors of salamander retina.J Neurosci. 2012 Dec 12;32(50):18112-23. doi: 10.1523/JNEUROSCI.1764-12.2012. J Neurosci. 2012. PMID: 23238726 Free PMC article.
-
Synaptojanin1 is required for temporal fidelity of synaptic transmission in hair cells.PLoS Genet. 2009 May;5(5):e1000480. doi: 10.1371/journal.pgen.1000480. Epub 2009 May 8. PLoS Genet. 2009. PMID: 19424431 Free PMC article.
References
-
- Balkema G, Jones N, Chalifoux J, Mayer J (2003) Co-localization of focal adhesion proteins with the synaptic ribbon in the photoreceptor terminal. Soc Neurosci Abstr 29: 475.13.
-
- Ball SL, Powers PA, Shin HS, Morgans CW, Peachey NS, Gregg RG (2002) Role of the beta(2) subunit of voltage-dependent calcium channels in the retinal outer plexiform layer. Invest Ophthalmol Vis Sci 43: 1595-1603. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
