The fragile X protein controls microtubule-associated protein 1B translation and microtubule stability in brain neuron development
- PMID: 15475576
- PMCID: PMC524058
- DOI: 10.1073/pnas.0404995101
The fragile X protein controls microtubule-associated protein 1B translation and microtubule stability in brain neuron development
Abstract
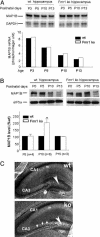
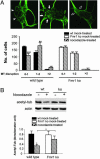
The fragile X mental retardation protein (FMRP) is a selective RNA-binding protein implicated in regulating translation of its mRNA ligands. The absence of FMRP results in fragile X syndrome, one of the leading causes of inherited mental retardation. Delayed dendritic spine maturation was found in fragile X mental retardation patients as well as in Fmr1 knockout (KO) mice, indicating the functional requirement of FMRP in synaptic development. However, the biochemical link between FMRP deficiency and the neuronal impairment during brain development has not been defined. How FMRP governs normal synapse development in the brain remains elusive. We report here that the developmentally programmed FMRP expression represses the translation of microtubule associated protein 1B (MAP1B) and is required for the accelerated decline of MAP1B during active synaptogenesis in neonatal brain development. The lack of FMRP results in misregulated MAP1B translation and delayed MAP1B decline in the Fmr1 KO brain. Furthermore, the aberrantly elevated MAP1B protein expression leads to abnormally increased microtubule stability in Fmr1 KO neurons. Together, these results indicate that FMRP plays critical roles in controlling cytoskeleton organization during neuronal development, and the abnormal microtubule dynamics is a conceivable underlying factor for the pathogenesis of fragile X mental retardation.
Figures



References
-
- Jin, P. & Warren, S. T. (2003) Trends Biochem. Sci. 28, 152-158. - PubMed
-
- O'Donnell, W. T. & Warren, S. T. (2002) Annu. Rev. Neurosci. 25, 315-338. - PubMed
-
- Bardoni, B. & Mandel, J. L. (2002) Curr. Opin. Genet. Dev. 12, 284-293. - PubMed
-
- Reiss, A. L., Aylward, E., Freund, L. S., Joshi, P. K. & Bryan, R. N. (1991) Ann. Neurol. 29, 26-32. - PubMed
-
- Irwin, S. A., Galvez, R. & Greenough, W. T. (2000) Cereb. Cortex 10, 1038-1044. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

