Proteomic analysis identifies that 14-3-3zeta interacts with beta-catenin and facilitates its activation by Akt
- PMID: 15492215
- PMCID: PMC524456
- DOI: 10.1073/pnas.0406499101
Proteomic analysis identifies that 14-3-3zeta interacts with beta-catenin and facilitates its activation by Akt
Abstract
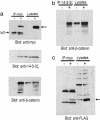
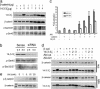
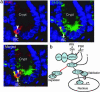
beta-Catenin is a central effector of Wnt signaling in embryonic and stem cell development and in tumorigenesis. Here, through a mass spectrometric analysis of a beta-catenin protein complex, we identified 12 proteins as putative beta-catenin interactors. We show that one of them, 14-3-3zeta, enhances beta-catenin-dependent transcription by maintaining a high level of beta-catenin protein in the cytoplasm. More importantly, 14-3-3zeta facilitates activation of beta-catenin by the survival kinase Akt and colocalizes with activated Akt in intestinal stem cells. We propose that Akt phosphorylates beta-catenin, which results in 14-3-3zeta binding and stabilization of beta-catenin, and these interactions may be involved in stem cell development.
Figures

Similar articles
-
Stabilization of beta-catenin by a Wnt-independent mechanism regulates cardiomyocyte growth.Proc Natl Acad Sci U S A. 2003 Apr 15;100(8):4610-5. doi: 10.1073/pnas.0835895100. Epub 2003 Mar 31. Proc Natl Acad Sci U S A. 2003. PMID: 12668767 Free PMC article.
-
Regulation of the versican promoter by the beta-catenin-T-cell factor complex in vascular smooth muscle cells.J Biol Chem. 2005 Apr 1;280(13):13019-28. doi: 10.1074/jbc.M411766200. Epub 2005 Jan 24. J Biol Chem. 2005. PMID: 15668231
-
Regulation of lymphoid enhancer factor 1/T-cell factor by mitogen-activated protein kinase-related Nemo-like kinase-dependent phosphorylation in Wnt/beta-catenin signaling.Mol Cell Biol. 2003 Feb;23(4):1379-89. doi: 10.1128/MCB.23.4.1379-1389.2003. Mol Cell Biol. 2003. PMID: 12556497 Free PMC article.
-
Phosphorylation of beta-catenin by AKT promotes beta-catenin transcriptional activity.J Biol Chem. 2007 Apr 13;282(15):11221-9. doi: 10.1074/jbc.M611871200. Epub 2007 Feb 7. J Biol Chem. 2007. PMID: 17287208 Free PMC article.
-
Signaling through beta-catenin and Lef/Tcf.Cell Mol Life Sci. 1999 Oct 30;56(5-6):523-37. doi: 10.1007/s000180050449. Cell Mol Life Sci. 1999. PMID: 11212302 Free PMC article. Review.
Cited by
-
PTEN-deficient intestinal stem cells initiate intestinal polyposis.Nat Genet. 2007 Feb;39(2):189-98. doi: 10.1038/ng1928. Epub 2007 Jan 21. Nat Genet. 2007. PMID: 17237784 Free PMC article.
-
Stress Can Induce Bovine Alpha-Herpesvirus 1 (BoHV-1) Reactivation from Latency.Viruses. 2024 Oct 27;16(11):1675. doi: 10.3390/v16111675. Viruses. 2024. PMID: 39599791 Free PMC article. Review.
-
Adenosine A2A receptor (A2AR) activation triggers Akt signaling and enhances nuclear localization of β-catenin in osteoblasts.FASEB J. 2019 Jun;33(6):7555-7562. doi: 10.1096/fj.201900014R. Epub 2019 Mar 13. FASEB J. 2019. PMID: 30866652 Free PMC article.
-
Wnt/beta-catenin signaling and small molecule inhibitors.Curr Pharm Des. 2013;19(4):634-64. doi: 10.2174/138161213804581837. Curr Pharm Des. 2013. PMID: 23016862 Free PMC article. Review.
-
Legionella pneumophila regulates host cell motility by targeting Phldb2 with a 14-3-3ζ-dependent protease effector.Elife. 2022 Feb 17;11:e73220. doi: 10.7554/eLife.73220. Elife. 2022. PMID: 35175192 Free PMC article.
References
-
- Miller, J. R. & Moon, R. T. (1996) Genes Dev. 10, 2527-2539. - PubMed
-
- Cadigan, K. M. & Nusse, R. (1997) Genes Dev. 11, 3286-3305. - PubMed
-
- Bienz, M. & Clevers, H. (2000) Cell 103, 311-320. - PubMed
-
- Zhurinsky, J., Shtutman, M. & Ben-Ze'ev, A. (2000) J. Cell Sci. 113, 3127-3139. - PubMed
-
- Peifer, M., Pai, L. M. & Casey, M. (1994) Dev. Biol. 166, 543-556. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

