Cross correlation by neurons of the medial superior olive: a reexamination
- PMID: 15492883
- PMCID: PMC2504554
- DOI: 10.1007/s10162-004-4027-4
Cross correlation by neurons of the medial superior olive: a reexamination
Abstract
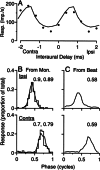
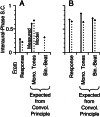
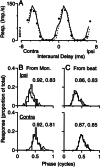
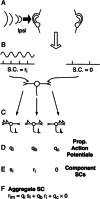
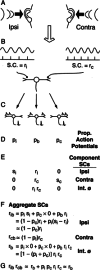
Initial analysis of interaural temporal disparities (ITDs), a cue for sound localization, occurs in the superior olivary complex. The medial superior olive (MSO) receives excitatory input from the left and right cochlear nuclei. Its neurons are believed to be coincidence detectors, discharging when input arrives simultaneously from the two sides. Many current psychophysical models assume a strict version of coincidence, in which neurons of the MSO cross correlate their left and right inputs. However, there have been few tests of this assumption. Here we examine data derived from two earlier studies of the MSO and compare the responses to the output of a computational model. We find that the MSO is not an ideal cross correlator. Ideal cross correlation implies a strict relationship between the precision of phase-locking of the inputs and the range of ITDs to which a neuron responds. This relationship does not appear to be met. Instead, the modeling implies that a neuron responds over a wider range of ITDs than expected from the inferred precision of phase-locking of the inputs. The responses are more consistent with a scheme in which the neuron can also be activated by the input from one side alone. Such activation degrades the tuning of neurons in the MSO to ITDs.
Figures




Similar articles
-
Directional hearing by linear summation of binaural inputs at the medial superior olive.Neuron. 2013 Jun 5;78(5):936-48. doi: 10.1016/j.neuron.2013.04.028. Neuron. 2013. PMID: 23764292 Free PMC article.
-
Envelope coding in the lateral superior olive. II. Characteristic delays and comparison with responses in the medial superior olive.J Neurophysiol. 1996 Oct;76(4):2137-56. doi: 10.1152/jn.1996.76.4.2137. J Neurophysiol. 1996. PMID: 8899590
-
Frequency-dependent interaural delays in the medial superior olive: implications for interaural cochlear delays.J Neurophysiol. 2011 Oct;106(4):1985-99. doi: 10.1152/jn.00131.2011. Epub 2011 Jul 20. J Neurophysiol. 2011. PMID: 21775710
-
The lateral superior olive: a functional role in sound source localization.Neuroscientist. 2003 Apr;9(2):127-43. doi: 10.1177/1073858403252228. Neuroscientist. 2003. PMID: 12708617 Review.
-
The evolution of temporal processing in the medial superior olive, an auditory brainstem structure.Prog Neurobiol. 2000 Aug;61(6):581-610. doi: 10.1016/s0301-0082(99)00068-4. Prog Neurobiol. 2000. PMID: 10775798 Review.
Cited by
-
Models of brainstem responses to bilateral electrical stimulation.J Assoc Res Otolaryngol. 2009 Mar;10(1):91-110. doi: 10.1007/s10162-008-0141-z. Epub 2008 Oct 22. J Assoc Res Otolaryngol. 2009. PMID: 18941838 Free PMC article.
-
Neural and behavioral sensitivity to interaural time differences using amplitude modulated tones with mismatched carrier frequencies.J Assoc Res Otolaryngol. 2007 Sep;8(3):393-408. doi: 10.1007/s10162-007-0088-5. Epub 2007 Jul 27. J Assoc Res Otolaryngol. 2007. PMID: 17657543 Free PMC article.
-
Detection of interaural time differences in the alligator.J Neurosci. 2009 Jun 24;29(25):7978-90. doi: 10.1523/JNEUROSCI.6154-08.2009. J Neurosci. 2009. PMID: 19553438 Free PMC article.
-
Sound Localization Strategies in Three Predators.Brain Behav Evol. 2015 Sep;86(1):17-27. doi: 10.1159/000435946. Epub 2015 Sep 24. Brain Behav Evol. 2015. PMID: 26398572 Free PMC article. Review.
-
Population rate-coding predicts correctly that human sound localization depends on sound intensity.Elife. 2019 Oct 21;8:e47027. doi: 10.7554/eLife.47027. Elife. 2019. PMID: 31633481 Free PMC article.
References
-
- Batra R, Fitzpatrick DC. Processing of interaural temporal disparities in the medial division of the ventral nucleus of the lateral lemniscus. J. Neurophysiol. 2002;88:666–675. - PubMed
-
- Batra R, Kuwada S, Fitzpatrick DC. Sensitivity to interaural temporal disparities of low- and high-frequency neurons in the superior olivary complex: I. Heterogeneity of responses. J. Neurophysiol. 1997a;78:1222–1236. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

