The helical alanine controversy: an (Ala)6 insertion dramatically increases helicity
- PMID: 15493925
- PMCID: PMC3870188
- DOI: 10.1021/ja047265o
The helical alanine controversy: an (Ala)6 insertion dramatically increases helicity
Abstract
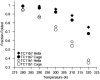
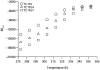
Employing chemical shift melts and hydrogen/deuterium exchange NMR techniques, we have determined the stabilization of the Trp-cage miniprotein due to multiple alanine insertions within the N-terminal alpha-helix. Alanine is shown to be uniquely helix-stabilizing and this stabilization is reflected in the global fold stability of the Trp-cage. The associated free energy change per alanine can be utilized to calculate the alanine propagation value. From the Lifson-Roig formulation, the calculated value (wAla = 1.6) is comparable to those obtained for short, solubilized, alanine-rich helices and is much larger than the values obtained by prior host-guest techniques or in N-terminally templated helices and peptides bearing long contiguous strings of alanines with no capping or solubilizing units present.
Copyright 2004 American Chemical Society
Figures
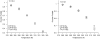


References
-
- Platzer KEB, Ananthanarayanan VS, Andreatta RH, Scheraga HA. Macromolecules. 1972;5:177–187.
- Scheraga HA. Pure Applied Chem. 1973;36:1–8.
- Wojcik J, Altmann KH, Scheraga HA. Biopolymers. 1990;30:121–134. - PubMed
-
- Scheraga HA, Vila JA, Ripoll DR. Biophys Chem. 2002;101-102:255–265. - PubMed
-
- Lifson S, Roig A. J Chem Phys. 1961;34:1963–1974.
- Doig AJ, Chakrabartty A, Klingler TM, Baldwin RL. Biochemistry. 1994;33:3396–3403. - PubMed
-
-
Helix propensities of different residues have been rationalized in terms of side-chain entropy differences between the random coil and helical state.6a–c Helicity increases for Xaa → Ala mutations can be attributed to a reduction in ΔSU. In addition, Ala's lack of a bulky side chain allows for stabilization of H-bonding interactions with water in the helical state.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

