Structural organization of mRNA complexes with major core mRNP protein YB-1
- PMID: 15494450
- PMCID: PMC524299
- DOI: 10.1093/nar/gkh889
Structural organization of mRNA complexes with major core mRNP protein YB-1
Abstract
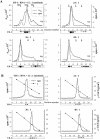
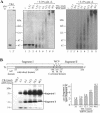
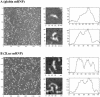
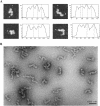
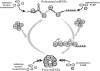
YB-1 is a universal major protein of cytoplasmic mRNPs, a member of the family of multifunctional cold shock domain proteins (CSD proteins). Depending on its amount on mRNA, YB-1 stimulates or inhibits mRNA translation. In this study, we have analyzed complexes formed in vitro at various YB-1 to mRNA ratios, including those typical for polysomal (translatable) and free (untranslatable) mRNPs. We have shown that at mRNA saturation with YB-1, this protein alone is sufficient to form mRNPs with the protein/RNA ratio and the sedimentation coefficient typical for natural mRNPs. These complexes are dynamic structures in which the protein can easily migrate from one mRNA molecule to another. Biochemical studies combined with atomic force microscopy and electron microscopy showed that mRNA-YB-1 complexes with a low YB-1/mRNA ratio typical for polysomal mRNPs are incompact; there, YB-1 binds to mRNA as a monomer with its both RNA-binding domains. At a high YB-1/mRNA ratio typical for untranslatable mRNPs, mRNA-bound YB-1 forms multimeric protein complexes where YB-1 binds to mRNA predominantly with its N-terminal part. A multimeric YB-1 comprises about twenty monomeric subunits; its molecular mass is about 700 kDa, and it packs a 600-700 nt mRNA segment on its surface.
Figures



Similar articles
-
Cytoplasmic mRNA-protein complexes of chicken muscle cells and their role in protein synthesis.Eur J Biochem. 1984 Jun 1;141(2):247-54. doi: 10.1111/j.1432-1033.1984.tb08184.x. Eur J Biochem. 1984. PMID: 6734598
-
Atomic force microscopy reveals binding of mRNA to microtubules mediated by two major mRNP proteins YB-1 and PABP.FEBS Lett. 2008 Aug 20;582(19):2875-81. doi: 10.1016/j.febslet.2008.07.019. Epub 2008 Jul 22. FEBS Lett. 2008. PMID: 18652827
-
An acidic protein, YBAP1, mediates the release of YB-1 from mRNA and relieves the translational repression activity of YB-1.Mol Cell Biol. 2005 Mar;25(5):1779-92. doi: 10.1128/MCB.25.5.1779-1792.2005. Mol Cell Biol. 2005. PMID: 15713634 Free PMC article.
-
Storage of messenger RNA in eukaryotes: envelopment with protein, translational barrier at 5' side, or conformational masking by 3' side?Mol Reprod Dev. 1994 May;38(1):107-17. doi: 10.1002/mrd.1080380117. Mol Reprod Dev. 1994. PMID: 7914085 Review.
-
Principles and properties of eukaryotic mRNPs.Mol Cell. 2014 May 22;54(4):547-58. doi: 10.1016/j.molcel.2014.04.033. Mol Cell. 2014. PMID: 24856220 Review.
Cited by
-
Formation of amyloid-like fibrils by Y-box binding protein 1 (YB-1) is mediated by its cold shock domain and modulated by disordered terminal domains.PLoS One. 2012;7(5):e36969. doi: 10.1371/journal.pone.0036969. Epub 2012 May 8. PLoS One. 2012. PMID: 22590640 Free PMC article.
-
A conserved stem loop motif in the 5'untranslated region regulates transforming growth factor-β(1) translation.PLoS One. 2010 Aug 26;5(8):e12283. doi: 10.1371/journal.pone.0012283. PLoS One. 2010. PMID: 20865036 Free PMC article.
-
Purification of nuclear poly(A)-binding protein Nab2 reveals association with the yeast transcriptome and a messenger ribonucleoprotein core structure.J Biol Chem. 2009 Dec 11;284(50):34911-7. doi: 10.1074/jbc.M109.062034. Epub 2009 Oct 19. J Biol Chem. 2009. PMID: 19840948 Free PMC article.
-
YB-1 transcription in the postnatal brain is regulated by a bHLH transcription factor Math2 through an E-box sequence in the 5'-UTR of the gene.Mol Cell Biochem. 2009 Jul;327(1-2):267-75. doi: 10.1007/s11010-009-0065-x. Epub 2009 Feb 19. Mol Cell Biochem. 2009. PMID: 19225866
-
Overexpression of Y-box binding protein-1 in cervical cancer and its association with the pathological response rate to chemoradiotherapy.Med Oncol. 2012 Sep;29(3):1992-7. doi: 10.1007/s12032-011-0062-2. Epub 2011 Sep 11. Med Oncol. 2012. PMID: 21909940
References
-
- Spirin A.S. (1996) Masked and translatable messenger ribonucleoproteins in higher eukaryores. In Hershey,J.W.B., Mathews,M.B. and Sonenberg,N. (eds), Translational Control. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, pp. 319–334.
-
- Samarina O.P. (1996) hnRNP particles. Bioessays, 18, 595–601.
-
- Dreyfuss G., Kim,V.N. and Kataoka,N. (2002) Messenger-RNA-binding proteins and the messages they carry. Nature Rev. Mol. Cell. Biol., 3, 195–205. - PubMed
-
- Kloc M., Zearfoss,N.R. and Etkin,L.D. (2002) Mechanisms of subcellular mRNA localization. Cell, 108, 533–544. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

