Cranial afferent glutamate heterosynaptically modulates GABA release onto second-order neurons via distinctly segregated metabotropic glutamate receptors
- PMID: 15496669
- PMCID: PMC6730106
- DOI: 10.1523/JNEUROSCI.1991-04.2004
Cranial afferent glutamate heterosynaptically modulates GABA release onto second-order neurons via distinctly segregated metabotropic glutamate receptors
Abstract
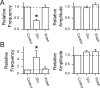
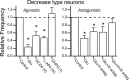
The balance between excitation and inhibition dictates central integration. Glutamatergic and GABAergic neurotransmission dominate this process. Cranial primary afferents enter the brainstem to release glutamate (Glu) onto second-order neurons within the caudal nucleus tractus solitarius (NTS) to initiate autonomic reflexes. The simplest pathways for these reflexes contain as few as two central neurons, but display robust frequency-dependent behavior. Within NTS, multiple metabotropic Glu receptors (mGluRs) are present, but their roles are poorly understood. Using synaptically discriminated second-order NTS neurons in brainstem slices and mechanically dissociated NTS neurons with intact boutons, we show that Glu differentially controls GABA release via distinct presynaptic mGluRs. In second-order NTS neurons recorded in slices, activation of primary afferents at frequencies as low as 10 shocks per second released sufficient Glu to alter rates of spontaneous IPSCs (sIPSCs). In both approaches, group I mGluRs increased GABA release in some neurons, but, on different neurons, group II and group III mGluRs decreased the sIPSC rate. mGluR actions were remarkably rapid, with onset and reversal beginning within 100 msec. In all cases, mGluR actions were exclusively presynaptic, and mGluRs did not alter postsynaptic properties in second-order neurons in either slices or isolated neurons. Tests with capsaicin and alphabeta-methylene ATP suggest that myelinated and unmyelinated afferent pathways engage both mGluR-GABA mechanisms. Afferent Glu spillover provides heterosynaptic cross talk with GABAergic inhibition in NTS. This process may critically shape the dynamic character and use dependence for cranial afferent transmission at the first stage of autonomic reflexes.
Figures

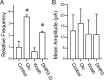
Similar articles
-
Heterosynaptic crosstalk: GABA-glutamate metabotropic receptors interactively control glutamate release in solitary tract nucleus.Neuroscience. 2011 Feb 3;174:1-9. doi: 10.1016/j.neuroscience.2010.11.053. Epub 2010 Dec 1. Neuroscience. 2011. PMID: 21129447 Free PMC article.
-
Glutamate suppresses GABA release via presynaptic metabotropic glutamate receptors at baroreceptor neurones in rats.J Physiol. 2005 Jan 15;562(Pt 2):535-51. doi: 10.1113/jphysiol.2004.076885. Epub 2004 Nov 11. J Physiol. 2005. PMID: 15539399 Free PMC article.
-
Synaptic transmission in nucleus tractus solitarius is depressed by Group II and III but not Group I presynaptic metabotropic glutamate receptors in rats.J Physiol. 2002 Feb 1;538(Pt 3):773-86. doi: 10.1113/jphysiol.2001.012948. J Physiol. 2002. PMID: 11826164 Free PMC article.
-
Sensory afferent neurotransmission in caudal nucleus tractus solitarius--common denominators.Chem Senses. 1996 Jun;21(3):387-95. doi: 10.1093/chemse/21.3.387. Chem Senses. 1996. PMID: 8670718 Review.
-
Channeling satiation: a primer on the role of TRP channels in the control of glutamate release from vagal afferent neurons.Physiol Behav. 2014 Sep;136:179-84. doi: 10.1016/j.physbeh.2014.09.003. Epub 2014 Oct 5. Physiol Behav. 2014. PMID: 25290762 Free PMC article. Review.
Cited by
-
Peptide and lipid modulation of glutamatergic afferent synaptic transmission in the solitary tract nucleus.Front Neurosci. 2013 Jan 10;6:191. doi: 10.3389/fnins.2012.00191. eCollection 2012. Front Neurosci. 2013. PMID: 23335875 Free PMC article.
-
Disinhibition of the cardiac limb of the arterial baroreflex in rat: a role for metabotropic glutamate receptors in the nucleus tractus solitarii.J Physiol. 2006 Sep 15;575(Pt 3):727-38. doi: 10.1113/jphysiol.2006.112672. Epub 2006 Jun 29. J Physiol. 2006. PMID: 16809369 Free PMC article.
-
GABA(B) restrains release from singly-evoked GABA terminals.Neuroscience. 2011 Oct 13;193:54-62. doi: 10.1016/j.neuroscience.2011.07.042. Epub 2011 Jul 27. Neuroscience. 2011. PMID: 21820490 Free PMC article.
-
Pancreatic insulin and exocrine secretion are under the modulatory control of distinct subpopulations of vagal motoneurones in the rat.J Physiol. 2012 Aug 1;590(15):3611-22. doi: 10.1113/jphysiol.2012.234955. Epub 2012 Jun 18. J Physiol. 2012. PMID: 22711959 Free PMC article.
-
Differential organization of excitatory and inhibitory synapses within the rat dorsal vagal complex.Am J Physiol Gastrointest Liver Physiol. 2011 Jan;300(1):G21-32. doi: 10.1152/ajpgi.00363.2010. Epub 2010 Oct 14. Am J Physiol Gastrointest Liver Physiol. 2011. PMID: 20947702 Free PMC article.
References
-
- Andresen MC, Kunze DL (1994) Nucleus tractus solitarius: gateway to neural circulatory control. Annu Rev Physiol 56: 93-116. - PubMed
-
- Andresen MC, Yang M (1990) Non-NMDA receptors mediate sensory afferent synaptic transmission in medial nucleus tractus solitarius. Am J Physiol 259: H1307-H1311. - PubMed
-
- Andresen MC, Yang M (1995) Dynamics of sensory afferent synaptic transmission in aortic baroreceptor regions of nucleus tractus solitarius. J Neurophysiol 74: 1518-1528. - PubMed
-
- Andresen MC, Doyle MW, Jin Y-H, Bailey TW (2001) Cellular mechanisms of baroreceptor integration at the nucleus tractus solitarius. Ann NY Acad Sci 940: 132-141. - PubMed
-
- Antunes VR, Machado BH (2003) Antagonism of glutamatergic metabotropic receptors in the NTS of awake rats does not affect the gain of the baroreflex. Auton Neurosci 103: 65-71. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
