Induction of a parafacial rhythm generator by rhombomere 3 in the chick embryo
- PMID: 15496674
- PMCID: PMC6730091
- DOI: 10.1523/JNEUROSCI.2408-04.2004
Induction of a parafacial rhythm generator by rhombomere 3 in the chick embryo
Abstract
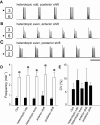
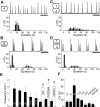
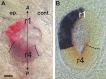
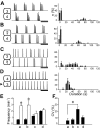
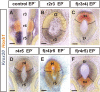
Observations of knock-out mice suggest that breathing at birth requires correct development of a specific hindbrain territory corresponding to rhombomeres (r) 3 and 4. Focusing on this territory, we examined the development of a neuronal rhythm generator in the chick embryo. We show that rhythmic activity in r4 is inducible after developmental stage 10 through interaction with r3. Although the nature of this interaction remains obscure, we find that the expression of Krox20, a segmentation gene responsible for specifying r3 and r5, is sufficient to endow other rhombomeres with the capacity to induce rhythmic activity in r4. Induction is robust, because it can be reproduced with r2 and r6 instead of r4 and with any hindbrain territory that normally expresses Krox20 (r3, r5) or can be forced to do so (r1, r4). Interestingly, the interaction between r4 and r3/r5 that results in rhythm production can only take place through the anterior border of r4, revealing a heretofore unsuspected polarity in individual rhombomeres. The r4 rhythm generator appears to be homologous to a murine respiratory parafacial neuronal system developing in r4 under the control of Krox20 and Hoxa1. These results identify a late role for Krox20 at the onset of neurogenesis.
Figures
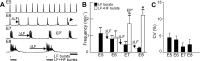

Similar articles
-
Early development of respiratory rhythm generation in mouse and chick.Respir Physiol Neurobiol. 2002 Jul;131(1-2):5-13. doi: 10.1016/s1569-9048(02)00033-2. Respir Physiol Neurobiol. 2002. PMID: 12106991 Review.
-
Rhombomere transplantation repatterns the segmental organization of cranial nerves and reveals cell-autonomous expression of a homeodomain protein.Development. 1993 Jan;117(1):105-17. doi: 10.1242/dev.117.1.105. Development. 1993. PMID: 7900983
-
Hindbrain patterning: FGFs regulate Krox20 and mafB/kr expression in the otic/preotic region.Development. 2000 Nov;127(22):4925-35. doi: 10.1242/dev.127.22.4925. Development. 2000. PMID: 11044406
-
Distinct roles of Hoxa2 and Krox20 in the development of rhythmic neural networks controlling inspiratory depth, respiratory frequency, and jaw opening.Neural Dev. 2007 Sep 26;2:19. doi: 10.1186/1749-8104-2-19. Neural Dev. 2007. PMID: 17897445 Free PMC article.
-
[Role of the Krox-20 gene in the development of rhombencephalon].C R Seances Soc Biol Fil. 1997;191(1):91-4. C R Seances Soc Biol Fil. 1997. PMID: 9181130 Review. French.
Cited by
-
Vertebrate Evolution Conserves Hindbrain Circuits despite Diverse Feeding and Breathing Modes.eNeuro. 2021 Apr 28;8(2):ENEURO.0435-20.2021. doi: 10.1523/ENEURO.0435-20.2021. Print 2021 Mar-Apr. eNeuro. 2021. PMID: 33707205 Free PMC article. Review.
-
Three brainstem areas involved in respiratory rhythm generation in bullfrogs.J Physiol. 2015 Jul 1;593(13):2941-54. doi: 10.1113/JP270380. Epub 2015 Jun 17. J Physiol. 2015. PMID: 25952282 Free PMC article.
-
Emergence of the pre-Bötzinger respiratory rhythm generator in the mouse embryo.J Neurosci. 2005 Apr 27;25(17):4307-18. doi: 10.1523/JNEUROSCI.0551-05.2005. J Neurosci. 2005. PMID: 15858057 Free PMC article.
-
Looking for inspiration: new perspectives on respiratory rhythm.Nat Rev Neurosci. 2006 Mar;7(3):232-42. doi: 10.1038/nrn1871. Nat Rev Neurosci. 2006. PMID: 16495944 Free PMC article. Review.
-
Development of Spontaneous Activity in the Avian Hindbrain.Front Neural Circuits. 2016 Aug 12;10:63. doi: 10.3389/fncir.2016.00063. eCollection 2016. Front Neural Circuits. 2016. PMID: 27570506 Free PMC article. Review.
References
-
- Abadie V, Champagnat J, Fortin G (2000) Branchiomotor activities in mouse embryo. NeuroReport 17: 141-145. - PubMed
-
- Barrow JR, Capecchi MR (1996) Targeted disruption of the Hoxb-2 locus in mice interferes with expression of Hoxb-1 and Hoxb-4. Development 122: 3817-3828. - PubMed
-
- Borday C, Abadie V, Chatonnet F, Thoby-Brisson M, Champagnat J, Fortin G (2003) Developmental molecular switches regulating breathing patterns in CNS. Respir Physiol Neurobiol 135: 121-132. - PubMed
-
- Brunet JF, Ghysen A (1999) Deconstructing cell determination: proneuronal genes and neuronal identity. BioEssays 21: 313-318. - PubMed
-
- Chatonnet F, Thoby-Brisson M, Abadie V, Dominguez del Toro E, Champagnat J, Fortin G (2002) Early development of respiratory rhythm generation in mouse and chick. Respir Physiol Neurobiol 131: 5-13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
