Pronounced cell death in the absence of NMDA receptors in the developing somatosensory thalamus
- PMID: 15496680
- PMCID: PMC6730102
- DOI: 10.1523/JNEUROSCI.3290-04.2004
Pronounced cell death in the absence of NMDA receptors in the developing somatosensory thalamus
Abstract
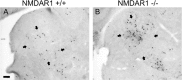
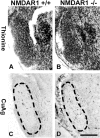
Genetic deletion of NMDA glutamate receptors disrupts development of whisker-related neuronal patterns in the somatosensory system. Independent studies have shown that NMDA receptor antagonists increase cell death among developing neurons. Here, we report that a dramatic feature of the developing somatosensory system in newborn NMDA receptor 1 (NMDAR1) knock-out mice is increased cell death in the ventrobasal nucleus (VB) of the thalamus. Sections were subject to terminal deoxynucleotidyl transferase dUTP nick end labeling staining for apoptotic DNA fragmentation, thionine staining for pyknotic nuclei, silver staining for degenerating cells, and immunostaining for caspase-3. All four methods demonstrated that deletion of NMDAR1 causes a large (on the order of threefold to fivefold) increase in cell death in the VB. The NMDA receptor antagonists dizocilpine maleate (MK-801) and phencyclidine also increase cell death in this structure. The onset of increased cell death in the VB in the absence of NMDA receptor function is approximately the time of birth, overlaps with naturally occurring cell death and synaptogenesis, and displays some anatomical specificity. For example, there was no increase in cell death in the hippocampus or neocortex of NMDAR1 knock-out mice at any of the time points examined: embryonic day 15.5 (E15.5), E17.5, and postnatal day 0. We also report a significant reduction in the size of the VB that is evident starting at E17.5. The results indicate that NMDA receptors play a major role in cell survival during naturally occurring cell death in the VB and demonstrate that cell death is a consideration in NMDA receptor knock-out studies.
Figures






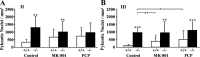


References
-
- Abel EL, Sokol RJ (1987) Incidence of fetal alcohol syndrome and economic impact of FAS-related anomalies. Drug Alcohol Depend 19: 51-70. - PubMed
-
- Abu-Qare AW, Abou-Donia MB (2001) Biomarkers of apoptosis: release of cytochrome c, activation of caspase-3, induction of 8-hydroxy-2′-deoxyguanosine, increased 3-nitrotyrosine, and alteration of p53 gene. J Toxicol Environ Health B Crit Rev 4: 313-332. - PubMed
-
- Aguayo LG, Albuquerque EX (1987) Phencyclidine blocks two potassium currents in spinal neurons in cell culture. Brain Res 436: 9-17. - PubMed
-
- Akesson E, Kjaeldgaard A, Samuelsson EB, Seiger A, Sundstrom E (2000) Ionotropic glutamate receptor expression in human spinal cord during first trimester development. Brain Res Dev Brain Res 119: 55-63. - PubMed
-
- Altman J, Bayer SA (1988) Development of the rat thalamus: IV. The intermediate lobule of the thalamic neuroepithelium, and the time and site of origin and settling pattern of neurons of the ventral nuclear complex. J Comp Neurol 284: 534-566. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
