Rapid, transient, and dose-dependent expression of hsp70 messenger RNA in the rat brain after morphine treatment
- PMID: 15497504
- PMCID: PMC1065297
- DOI: 10.1379/csc-42.1
Rapid, transient, and dose-dependent expression of hsp70 messenger RNA in the rat brain after morphine treatment
Abstract
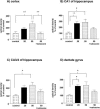
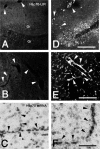
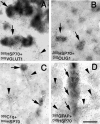
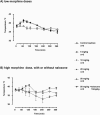
Induction of Hsp70 in the brain has been reported after intake of drugs of abuse like amphetamine and lysergic acid diethylamide. In this investigation, gene expression of Hsp70 and other heat shock genes in the rat brain was studied in response to morphine. Twenty milligrams per kilogram morphine intraperitoneally resulted in a marked induction of Hsp70 messenger RNA (mRNA) expression in the frontal cortex with a maximum increase of 13.2-fold after 2 hours. A moderate increase of Hsp27 mRNA expression (6.7-fold) could be observed after 4 hours, whereas mRNA expression of Hsp90 and of the constitutive Hsc70 did not exceed a mean factor of 1.8-fold during the 24 hours interval. The increase in Hsp70 mRNA was dose dependent, showing a significant elevation after doses ranging from 10 to 50 mg/kg morphine. In situ hybridization revealed enhanced Hsp70 mRNA expression mainly in cortical areas, in the hippocampus, in the paraventricular and supraoptic nuclei of the hypothalamus, in the locus coeruleus, as well in the pineal body. The double in situ hybridization technique revealed increased Hsp70 mRNA expression mainly in VGLUT1-positive neurons and to a lesser extent in olig1-positive oligodendroglia. Immunohistochemistry revealed a marked increase of Hsp70 protein in neuronal cells and blood vessels after 12 hours. In contrast to animal experiments, morphine did not increase Hsp70 mRNA expression in vitro in micro-opioid receptor (MOR1)-expressing human embryonic kidney 293 cells, suggesting no direct MOR1-mediated cellular effect. To exclude a body temperature-related morphine effect on Hsp70 mRNA expression, the temperature was recorded. Five to 20 mg/kg resulted in hyperthermia (maximum 40.6 degrees), whereas a high dose (50 mg/kg) that produced the highest mRNA induction, showed a clear hypothermia (minimum 37.2 degrees C). These findings argue against the possibility that Hsp70 induction by morphine is caused by its effect on body temperature. It may be speculated that increased expression of Hsp70 after morphine application protects brain structures against potentially hazardous effects of opiates.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
