Recognition of mycobacterial antigens delivered by genetically detoxified Bordetella pertussis adenylate cyclase by T cells from cattle with bovine tuberculosis
- PMID: 15501751
- PMCID: PMC523049
- DOI: 10.1128/IAI.72.11.6255-6261.2004
Recognition of mycobacterial antigens delivered by genetically detoxified Bordetella pertussis adenylate cyclase by T cells from cattle with bovine tuberculosis
Abstract
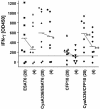
The exponential increase in the incidence of tuberculosis in cattle over the last two decades in the British national herd constitutes a significant economic problem. Therefore, research efforts are under way to develop cattle tuberculosis vaccines and specific diagnostic reagents to allow the distinction of vaccinated from infected animals. Mycobacterial antigens like ESAT-6 and CFP10 allow this distinction. This study investigates whether fusion protein of ESAT-6 or CFP10 with genetically detoxified Bordetella pertussis adenylate cyclase (CyaA) are recognized by Mycobacterium bovis-infected cattle more effectively than conventional recombinant proteins are, thus enhancing sensitivity or reducing the amount of antigens required. By measuring the frequencies of gamma interferon (IFN-gamma)-producing cells, we were able to show that the presentation of CFP10 as a CyaA fusion protein enhanced the molecular efficiency of its recognition 20-fold, while the recognition of ESAT-6 was not improved by CyaA delivery. Furthermore, in the whole-blood IFN-gamma test currently used in the field, the delivery of CFP10 and ESAT-6 by fusion to CyaA increased the amount of IFN-gamma produced and hence the proportion of infected animals responding to CFP10. The improved T-cell recognition of CyaA336/CFP10 was found to be dependent upon interaction with CD11b. In addition, presentation of CyaA336/CFP10 to CD4+ T cells was chloroquine sensitive, and CFP10 delivery by CyaA resulted in its accelerated presentation to T cells. In conclusion, the use of CyaA fusion proteins with ESAT-6 and CFP10 has the potential to improve the sensitivity of immunodiagnostic tests detecting bovine tuberculosis in cattle.
Figures




References
-
- Accapezzato, D., R. Nisini, M. Paroli, G. Bruno, F. Bonino, M. Houghton, and V. Barnaba. 1998. Generation of an MHC class II-restricted T cell epitope by extracellular processing of hepatitis delta antigen. J. Immunol. 160:5262-5266. - PubMed
-
- Behr, M. A., M. A. Wilson, W. P. Gill, H. Salamon, G. K. Schoolnik, S. Rane, and P. M. Small. 1999. Comparative genomics of BCG vaccines by whole-genome DNA microarray. Science 284:1520-1523. - PubMed
-
- Buddle, B. M., A. R. McCarthy, T. J. Ryan, J. M. Pollock, H. M. Vordermeier, R. G. Hewinson, P. Andersen, and G. W. de Lisle. 2003. Use of mycobacterial peptides and recombinant proteins for the diagnosis of bovine tuberculosis in skin test-positive cattle. Vet. Rec. 153:615-620. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

