Respiratory hydrogen use by Salmonella enterica serovar Typhimurium is essential for virulence
- PMID: 15501756
- PMCID: PMC523013
- DOI: 10.1128/IAI.72.11.6294-6299.2004
Respiratory hydrogen use by Salmonella enterica serovar Typhimurium is essential for virulence
Abstract
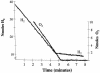
Based on available annotated gene sequence information, the enteric pathogen salmonella, like other enteric bacteria, contains three putative membrane-associated H2-using hydrogenase enzymes. These enzymes split molecular H2, releasing low-potential electrons that are used to reduce quinone or heme-containing components of the respiratory chain. Here we show that each of the three distinct membrane-associated hydrogenases of Salmonella enterica serovar Typhimurium is coupled to a respiratory pathway that uses oxygen as the terminal electron acceptor. Cells grown in a blood-based medium expressed four times the amount of hydrogenase (H2 oxidation) activity that cells grown on Luria Bertani medium did. Cells suspended in phosphate-buffered saline consumed 2 mol of H2 per mol of O2 used in the H2-O2 respiratory pathway, and the activity was inhibited by the respiration inhibitor cyanide. Molecular hydrogen levels averaging over 40 microM were measured in organs (i.e., livers and spleens) of live mice, and levels within the intestinal tract (the presumed origin of the gas) were four times greater than this. The half-saturation affinity of S. enterica serovar Typhimurium for H2 is only 2.1 microM, so it is expected that H2-utilizing hydrogenase enzymes are saturated with the reducing substrate in vivo. All three hydrogenase enzymes contribute to the virulence of the bacterium in a typhoid fever-mouse model, based on results from strains with mutations in each of the three hydrogenase genes. The introduced mutations are nonpolar, and growth of the mutant strains was like that of the parent strain. The combined removal of all three hydrogenases resulted in a strain that is avirulent and (in contrast to the parent strain) one that is unable to invade liver or spleen tissue. The introduction of one of the hydrogenase genes into the triple mutant strain on a low-copy-number plasmid resulted in a strain that was able to both oxidize H2 and cause morbidity in mice within 11 days of inoculation; therefore, the avirulent phenotype of the triple mutant is not due to an unknown spurious mutation. We conclude that H2 utilization in a respiratory fashion is required for energy production to permit salmonella growth and subsequent virulence during infection.
Figures

References
-
- Böck, A., and G. Sawers. 2002. Fermentation, p. 262-282. In F. C. Neidhardt et al. (ed.), Escherichia coli and Salmonella: cellular and molecular biology, 2nd ed., vol. 1. ASM Press, Washington, D.C.
-
- Maier, R. J. 2003. Availability and use of molecular hydrogen as an energy substrate for Helicobacter species. Microbes Infect. 12:1159-1163. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

