Streptococcus mutans surface alpha-enolase binds salivary mucin MG2 and human plasminogen
- PMID: 15501816
- PMCID: PMC523000
- DOI: 10.1128/IAI.72.11.6748-6752.2004
Streptococcus mutans surface alpha-enolase binds salivary mucin MG2 and human plasminogen
Abstract
Matrix-assisted laser desorption ionization-time of flight mass spectrometry analysis identified enolase as a cell surface component of Streptococcus mutans, which was confirmed by enzyme-linked immunosorbent assay, Western blotting, and transmission electron microscopy. Surface enolase was demonstrated to bind to human plasminogen and salivary mucin MG2. The results suggested a role for enolase in S. mutans attachment, clearance, or breach of the bloodstream barrier.
Figures
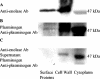

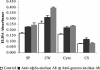
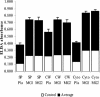
Similar articles
-
Cloning and characterization of an alpha-enolase of the oral pathogen Streptococcus mutans that binds human plasminogen.Biochem Biophys Res Commun. 2007 Dec 28;364(4):924-9. doi: 10.1016/j.bbrc.2007.10.098. Epub 2007 Oct 25. Biochem Biophys Res Commun. 2007. PMID: 17964283 Free PMC article.
-
Isolation of human salivary mucin MG2 by a novel method and characterization of its interactions with oral bacteria.Arch Biochem Biophys. 1999 Apr 15;364(2):286-93. doi: 10.1006/abbi.1999.1141. Arch Biochem Biophys. 1999. PMID: 10190986
-
Echinostoma caproni: identification of enolase in excretory/secretory products, molecular cloning, and functional expression.Exp Parasitol. 2007 Sep;117(1):57-64. doi: 10.1016/j.exppara.2007.03.011. Epub 2007 Mar 27. Exp Parasitol. 2007. PMID: 17462631
-
[Enolase on the surface of prockaryotic and eukaryotic cells is a receptor for human plasminogen].Postepy Hig Med Dosw (Online). 2007 Nov 15;61:672-82. Postepy Hig Med Dosw (Online). 2007. PMID: 18033204 Review. Polish.
-
[Enolase and parasitic infection].Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 2014 Aug;26(4):445-8. Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 2014. PMID: 25434150 Review. Chinese.
Cited by
-
Phenotypic characterization of Streptococcus pneumoniae biofilm development.J Bacteriol. 2006 Apr;188(7):2325-35. doi: 10.1128/JB.188.7.2325-2335.2006. J Bacteriol. 2006. PMID: 16547018 Free PMC article.
-
Plasminogen alleles influence susceptibility to invasive aspergillosis.PLoS Genet. 2008 Jun 20;4(6):e1000101. doi: 10.1371/journal.pgen.1000101. PLoS Genet. 2008. PMID: 18566672 Free PMC article.
-
MUC7 Level As A New Saliva Risk Factor For Dental Caries In Adult Patients.Int J Med Sci. 2019 Jan 1;16(2):241-246. doi: 10.7150/ijms.29027. eCollection 2019. Int J Med Sci. 2019. PMID: 30745804 Free PMC article.
-
Safety assessment of probiotics for human use.Gut Microbes. 2010 May-Jun;1(3):164-85. doi: 10.4161/gmic.1.3.12127. Epub 2010 Mar 4. Gut Microbes. 2010. PMID: 21327023 Free PMC article. Review.
-
From Normal Flora to Brain Abscesses: A Review of Streptococcus intermedius.Front Microbiol. 2020 May 7;11:826. doi: 10.3389/fmicb.2020.00826. eCollection 2020. Front Microbiol. 2020. PMID: 32457718 Free PMC article. Review.
References
-
- Ajdic, D., W. M. McShan, R. E. McLaughlin, G. Savic, J. Chang, M. B. Carson, C. Primeaux, R. Tian, S. Kenton, H. Jia, S. Lin, Y. Qian, S. Li, H. Zhu, F. Najar, H. Lai, J. White, B. A. Joe, and J. J. Ferretti. 2002. Genomic sequence of Streptococcus mutans UA159, a cariogenic dental pathogen. Proc. Natl. Acad. Sci. USA 99:14434-14439. - PMC - PubMed
-
- Banas, A. J. 2004. Virulence properties of Streptococcus mutans. Front. Biosci. 9:1267-1277. - PubMed
-
- Bergmann, S., M. Rohde, G. S. Chhatwal, and S. Hammerschmidt. 2001. α-Enolase of Streptococcus pneumoniae is a plasminogen-binding protein displayed on the bacterial cell surface. Mol. Microbiol. 40:1273-1287. - PubMed
-
- Bottalico, L. A., N. C. Kendrick, A. Keller, Y. Li, and I. Tabas. 1993. Cholesteryl ester loading of mouse peritoneal macrophages is associated with changes in expression or modification of specific cellular proteins, including increase in an α-enolase isoform. Arterioscl. Thromb. 13:264-275. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

