Calcium release from presynaptic ryanodine-sensitive stores is required for long-term depression at hippocampal CA3-CA3 pyramidal neuron synapses
- PMID: 15509748
- PMCID: PMC6730140
- DOI: 10.1523/JNEUROSCI.5583-03.2004
Calcium release from presynaptic ryanodine-sensitive stores is required for long-term depression at hippocampal CA3-CA3 pyramidal neuron synapses
Abstract
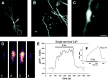
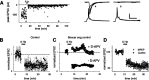
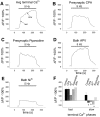
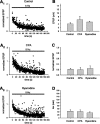
Although Ca2+ release from internal stores has been proposed to be important for the induction of long-term synaptic plasticity, the importance of Ca2+ stores localized in presynaptic terminals remains unclear. Here, we have selectively applied pharmacological antagonists to either the presynaptic or postsynaptic cell in paired whole-cell recordings from hippocampal CA3 pyramidal neurons in slice culture. We demonstrate directly the necessary role of presynaptic, but not postsynaptic, ryanodine-sensitive Ca2+ stores in the induction of NMDA receptor (NMDAR)-dependent long-term depression (LTD). Using two-photon laser scanning microscopy, we further find that release from the ryanodine-sensitive stores during prolonged synaptic stimulation generates a slowly rising Ca2+ signal in the presynaptic terminal that is required for the induction of LTD. Moreover, this form of LTD has a significant presynaptic component of expression because it causes a marked decrease in the rate of release from CA3 neuron presynaptic terminals of FM 1-43, a fluorescent probe of synaptic vesicle cycling. Thus, Ca2+ release from presynaptic ryanodine-sensitive stores is critical in the induction of a presynaptic component of NMDAR-dependent LTD.
Figures




Similar articles
-
Depolarization-induced long-term depression at hippocampal mossy fiber-CA3 pyramidal neuron synapses.J Neurosci. 2003 Oct 29;23(30):9786-95. doi: 10.1523/JNEUROSCI.23-30-09786.2003. J Neurosci. 2003. PMID: 14586006 Free PMC article.
-
Activation of presynaptic and postsynaptic ryanodine-sensitive calcium stores is required for the induction of long-term depression at GABAergic synapses in the neonatal rat hippocampus.J Neurosci. 2000 Sep 1;20(17):RC94. doi: 10.1523/JNEUROSCI.20-17-j0002.2000. J Neurosci. 2000. PMID: 10952733 Free PMC article.
-
Induction of hippocampal long-term depression requires release of Ca2+ from separate presynaptic and postsynaptic intracellular stores.J Neurosci. 1996 Oct 1;16(19):5951-60. doi: 10.1523/JNEUROSCI.16-19-05951.1996. J Neurosci. 1996. PMID: 8815877 Free PMC article.
-
Role of presynaptic metabotropic glutamate receptors in the induction of long-term synaptic plasticity of vesicular release.Neuropharmacology. 2013 Mar;66:31-9. doi: 10.1016/j.neuropharm.2012.05.004. Epub 2012 May 22. Neuropharmacology. 2013. PMID: 22626985 Free PMC article. Review.
-
Presence and functional significance of presynaptic ryanodine receptors.Prog Neurobiol. 2003 Apr;69(6):391-418. doi: 10.1016/s0301-0082(03)00053-4. Prog Neurobiol. 2003. PMID: 12880633 Review.
Cited by
-
Organelle-Specific Sensors for Monitoring Ca2+ Dynamics in Neurons.Front Synaptic Neurosci. 2016 Sep 16;8:29. doi: 10.3389/fnsyn.2016.00029. eCollection 2016. Front Synaptic Neurosci. 2016. PMID: 27695411 Free PMC article. Review.
-
Roles and Sources of Calcium in Synaptic Exocytosis.Adv Neurobiol. 2023;33:139-170. doi: 10.1007/978-3-031-34229-5_6. Adv Neurobiol. 2023. PMID: 37615866
-
Two coincidence detectors for spike timing-dependent plasticity in somatosensory cortex.J Neurosci. 2006 Apr 19;26(16):4166-77. doi: 10.1523/JNEUROSCI.0176-06.2006. J Neurosci. 2006. PMID: 16624937 Free PMC article.
-
CASK regulates CaMKII autophosphorylation in neuronal growth, calcium signaling, and learning.Front Mol Neurosci. 2013 Sep 11;6:27. doi: 10.3389/fnmol.2013.00027. eCollection 2013. Front Mol Neurosci. 2013. PMID: 24062638 Free PMC article.
-
Cellular signal mechanisms of reward-related plasticity in the hippocampus.Neural Plast. 2012;2012:945373. doi: 10.1155/2012/945373. Epub 2012 Nov 13. Neural Plast. 2012. PMID: 23213575 Free PMC article. Review.
References
-
- Arancio O, Kiebler M, Lee CJ, Lev-Ram V, Tsien RY, Kandel ER, Hawkins RD (1996) Nitric oxide acts directly in the presynaptic neuron to produce long-term potentiation in cultured hippocampal neurons. Cell 87: 1025-1035. - PubMed
-
- Ascher-Landsberg J, Saunders T, Elovitz M, Phillippe M (1999) The effects of 2-aminoethoxydiphenyl borate, a novel inositol 1,4,5-trisphosphate receptor modulator on myometrial contractions. Biochem Biophys Res Commun 264: 979-982. - PubMed
-
- Babcock DF, Hille B (1998) Mitochondrial oversight of cellular Ca2+ signaling. Curr Opin Neurobiol 8: 398-404. - PubMed
-
- Berridge MJ (1998) Neuronal calcium signaling. Neuron 21: 13-26. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
