The mitochondrial RNA polymerase contributes critically to promoter specificity in mammalian cells
- PMID: 15526033
- PMCID: PMC533051
- DOI: 10.1038/sj.emboj.7600465
The mitochondrial RNA polymerase contributes critically to promoter specificity in mammalian cells
Abstract
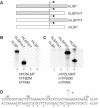
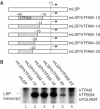
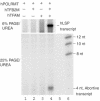
Initiation of transcription in mammalian mitochondria depends on three proteins: mitochondrial RNA polymerase (POLRMT), mitochondrial transcription factor A (TFAM) and mitochondrial transcription factor B2 (TFB2M). We show here that the recombinant mouse and human transcription machineries are unable to initiate transcription in vitro from the heterologous light-strand promoter (LSP) of mitochondrial DNA. This species specificity is dependent on the interaction of TFAM and POLRMT with specific distal and proximal promoter elements. A sequence element localized from position -1 to -2 relative to the transcription start site in LSP functionally interacts with POLRMT. The POLRMT/TFB2M heterodimer is unable to interact with promoter elements and initiate even abortive transcription in the absence of TFAM. TFAM is thus an integral part of the mammalian transcription machinery, and we propose that TFAM induces a structural change of the promoter that is required for POLRMT-dependent promoter recognition.
Figures





References
-
- Chang DD, Clayton DA (1984) Precise identification of individual promoters for transcription of each strand of human mitochondrial DNA. Cell 36: 635–643 - PubMed
-
- Cheetham GM, Steitz TA (2000) Insights into transcription: structure and function of single-subunit DNA-dependent RNA polymerases. Curr Opin Struct Biol 10: 117–123 - PubMed
-
- Clayton DA (1991) Replication and transcription of vertebrate mitochondrial DNA. Annu Rev Cell Biol 7: 453–478 - PubMed
-
- Dairaghi DJ, Shadel GS, Clayton DA (1995a) Addition of a 29 residue carboxyl-terminal tail converts a simple HMG box-containing protein into a transcriptional activator. J Mol Biol 249: 11–28 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

