Glutamine, glutamate, and alpha-glucosylglycerate are the major osmotic solutes accumulated by Erwinia chrysanthemi strain 3937
- PMID: 15528516
- PMCID: PMC525223
- DOI: 10.1128/AEM.70.11.6535-6541.2004
Glutamine, glutamate, and alpha-glucosylglycerate are the major osmotic solutes accumulated by Erwinia chrysanthemi strain 3937
Abstract
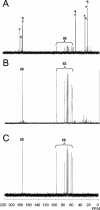
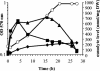
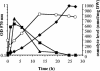
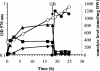
Erwinia chrysanthemi is a phytopathogenic soil enterobacterium closely related to Escherichia coli. Both species respond to hyperosmotic pressure and to external added osmoprotectants in a similar way. Unexpectedly, the pools of endogenous osmolytes show different compositions. Instead of the commonly accumulated glutamate and trehalose, E. chrysanthemi strain 3937 promotes the accumulation of glutamine and alpha-glucosylglycerate, which is a new osmolyte for enterobacteria, together with glutamine. The amounts of the three osmolytes increased with medium osmolarity and were reduced when betaine was provided in the growth medium. Both glutamine and glutamate showed a high rate of turnover, whereas glucosylglycerate stayed stable. In addition, the balance between the osmolytes depended on the osmolality of the medium. Glucosylglycerate and glutamate were the major intracellular compounds in low salt concentrations, whereas glutamine predominated at higher concentrations. Interestingly, the ammonium content of the medium also influenced the pool of osmolytes. During bacterial growth with 1 mM ammonium in stressing conditions, more glucosylglycerate accumulated by far than the other organic solutes. Glucosylglycerate synthesis has been described in some halophilic archaea and bacteria but not as a dominant osmolyte, and its role as an osmolyte in Erwinia chrysanthemi 3937 shows that nonhalophilic bacteria can also use ionic osmolytes.
Figures


References
-
- Back, D. M., and P. L. Polavarapu. 1983. Fourier-transform infrared spectroscopy of sugars. Structural changes in aqueous solutions. Carbohydr. Res. 121:308-311. - PubMed
-
- Booth, I. R., and C. F. Higgins. 1990. Enteric bacteria and osmotic stress: intracellular potassium glutamate as a secondary signal of osmotic stress? FEMS Microbiol. Rev. 75:239-246. - PubMed
-
- Bourot, S., O. Sire, A. Trautwetter, T. Touze, L. F. Wu, C. Blanco, and T. Bernard. 2000. Glycine betaine-assisted protein folding in a lysA mutant of Escherichia coli. J. Biol. Chem. 275:1050-1056. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

