Central localization of plasticity involved in appetitive conditioning in Lymnaea
- PMID: 15537733
- PMCID: PMC534707
- DOI: 10.1101/lm.77004
Central localization of plasticity involved in appetitive conditioning in Lymnaea
Abstract
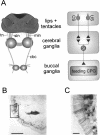
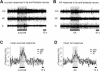
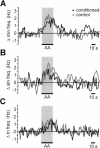
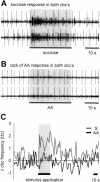
Learning to associate a conditioned (CS) and unconditioned stimulus (US) results in changes in the processing of CS information. Here, we address directly the question whether chemical appetitive conditioning of Lymnaea feeding behavior involves changes in the peripheral and/or central processing of the CS by using extracellular recording techniques to monitor neuronal activity at two stages of the sensory processing pathway. Our data show that appetitive conditioning does not affect significantly the overall CS response of afferent nerves connecting chemosensory structures in the lips and tentacles to the central nervous system (CNS). In contrast, neuronal output from the cerebral ganglia, which represent the first central processing stage for chemosensory information, is enhanced significantly in response to the CS after appetitive conditioning. This demonstrates that chemical appetitive conditioning in Lymnaea affects the central, but not the peripheral processing of chemosensory information. It also identifies the cerebral ganglia of Lymnaea as an important site for neuronal plasticity and forms the basis for detailed cellular studies of neuronal plasticity.
Figures

Similar articles
-
Selective expression of electrical correlates of differential appetitive classical conditioning in a feeding network.J Neurophysiol. 2001 Jan;85(1):89-97. doi: 10.1152/jn.2001.85.1.89. J Neurophysiol. 2001. PMID: 11152709
-
Effects of age on feeding behavior and chemosensory processing in the pond snail, Lymnaea stagnalis.Neurobiol Aging. 2006 Dec;27(12):1880-91. doi: 10.1016/j.neurobiolaging.2005.09.040. Epub 2005 Nov 8. Neurobiol Aging. 2006. PMID: 16280186
-
Cellular analysis of appetitive learning in invertebrates.Acta Biol Hung. 1999;50(1-3):117-29. Acta Biol Hung. 1999. PMID: 10574434
-
Pavlovian conditioning of Hermissenda: current cellular, molecular, and circuit perspectives.Learn Mem. 2004 May-Jun;11(3):229-38. doi: 10.1101/lm.70704. Learn Mem. 2004. PMID: 15169851 Review.
-
Feeding behavior of Aplysia: a model system for comparing cellular mechanisms of classical and operant conditioning.Learn Mem. 2006 Nov-Dec;13(6):669-80. doi: 10.1101/lm.339206. Learn Mem. 2006. PMID: 17142299 Review.
Cited by
-
What can we teach Lymnaea and what can Lymnaea teach us?Biol Rev Camb Philos Soc. 2021 Aug;96(4):1590-1602. doi: 10.1111/brv.12716. Epub 2021 Apr 6. Biol Rev Camb Philos Soc. 2021. PMID: 33821539 Free PMC article. Review.
-
A CREB2-targeting microRNA is required for long-term memory after single-trial learning.Sci Rep. 2018 Mar 2;8(1):3950. doi: 10.1038/s41598-018-22278-w. Sci Rep. 2018. PMID: 29500383 Free PMC article.
-
Plasticity of peptidergic innervation in healing rabbit medial collateral ligament.Can J Surg. 2008 Jun;51(3):167-72. Can J Surg. 2008. PMID: 18682794 Free PMC article.
-
pT305-CaMKII stabilizes a learning-induced increase in AMPA receptors for ongoing memory consolidation after classical conditioning.Nat Commun. 2014 May 30;5:3967. doi: 10.1038/ncomms4967. Nat Commun. 2014. PMID: 24875483 Free PMC article.
-
Effects of Aβ exposure on long-term associative memory and its neuronal mechanisms in a defined neuronal network.Sci Rep. 2015 May 29;5:10614. doi: 10.1038/srep10614. Sci Rep. 2015. PMID: 26024049 Free PMC article.
References
-
- Alexander Jr., J., Audesirk, T.E., and Audesirk, G.J. 1984. One-trial reward learning in the snail Lymnaea stagnalis. J. Neurobiol. 15: 67-72. - PubMed
-
- Alkon, D.L., Sakakibara, M., Forman, R., Harrigan, J., Lederhendler, I., and Farley, J. 1985. Reduction of two voltage-dependent K+ currents mediates retention of a learned association. Behav. Neural. Biol. 44: 278-300. - PubMed
-
- Benjamin, P.R. and Elliott, C.J.H. 1989. Snail feeding oscillator: The central pattern generator and its control by modulatory interneurons. In Neural and cellular oscillators (ed. J.W. Jacklet), pp. 173-214. Dekker, New York.
-
- Benjamin, P.R., Staras, K., and Kemenes, G. 2000. A systems approach to the cellular analysis of associative learning in the pond snail Lymnaea. Learn. Mem. 7: 124-131. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
