Regulation of the NMDA receptor complex and trafficking by activity-dependent phosphorylation of the NR2B subunit PDZ ligand
- PMID: 15537897
- PMCID: PMC6730169
- DOI: 10.1523/JNEUROSCI.0546-04.2004
Regulation of the NMDA receptor complex and trafficking by activity-dependent phosphorylation of the NR2B subunit PDZ ligand
Abstract
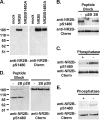
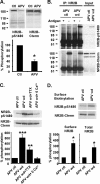
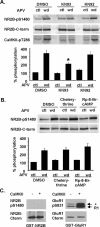
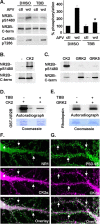
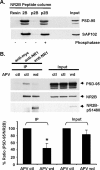
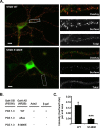
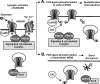
Interactions between NMDA receptors (NMDARs) and the PDZ [postsynaptic density-95 (PSD-95)/Discs large/zona occludens-1] domains of PSD-95/SAP90 (synapse-associated protein with a molecular weight of 90 kDa) family proteins play important roles in the synaptic targeting and signaling of NMDARs. However, little is known about the mechanisms that regulate these PDZ domain-mediated interactions. Here we show that casein kinase II (CK2) phosphorylates the serine residue (Ser1480) within the C-terminal PDZ ligand (IESDV) of the NR2B subunit of NMDAR in vitro and in vivo. Phosphorylation of Ser1480 disrupts the interaction of NR2B with the PDZ domains of PSD-95 and SAP102 and decreases surface NR2B expression in neurons. Interestingly, activity of the NMDAR and Ca2+/calmodulin-dependent protein kinase II regulates CK2 phosphorylation of Ser1480. Furthermore, CK2 colocalizes with NR1 and PSD-95 at synaptic sites. These results indicate that activity-dependent CK2 phosphorylation of the NR2B PDZ ligand regulates the interaction of NMDAR with PSD-95/SAP90 family proteins as well as surface NMDAR expression and may be a critical mechanism for modulating excitatory synaptic function and plasticity.
Figures

References
-
- Aoki C, Fujisawa S, Mahadomrongkul V, Shah PJ, Nader K, Erisir A (2003) NMDA receptor blockade in intact adult cortex increases trafficking of NR2A subunits into spines, postsynaptic densities, and axon terminals. Brain Res 963: 139-149. - PubMed
-
- Barria A, Malinow R (2002) Subunit-specific NMDA receptor trafficking to synapses. Neuron 35: 345-353. - PubMed
-
- Bayer KU, De Koninck P, Leonard AS, Hell JW, Schulman H (2001) Interaction with the NMDA receptor locks CaMKII in an active conformation. Nature 411: 801-805. - PubMed
-
- Beattie EC, Carroll RC, Yu X, Morishita W, Yasuda H, von Zastrow M, Malenka RC (2000) Regulation of AMPA receptor endocytosis by a signaling mechanism shared with LTD. Nat Neurosci 3: 1291-1300. - PubMed
-
- Blanquet PR (1998) Neurotrophin-induced activation of casein kinase 2 in rat hippocampal slices. Neuroscience 86: 739-749. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
