Peptidoglycan recognition protein (PGRP)-LE and PGRP-LC act synergistically in Drosophila immunity
- PMID: 15538387
- PMCID: PMC533052
- DOI: 10.1038/sj.emboj.7600466
Peptidoglycan recognition protein (PGRP)-LE and PGRP-LC act synergistically in Drosophila immunity
Abstract
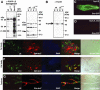
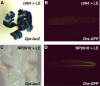
In innate immunity, pattern recognition molecules recognize cell wall components of microorganisms and activate subsequent immune responses, such as the induction of antimicrobial peptides and melanization in Drosophila. The diaminopimelic acid (DAP)-type peptidoglycan potently activates imd-dependent induction of antibacterial peptides. Peptidoglycan recognition protein (PGRP) family members act as pattern recognition molecules. PGRP-LC loss-of-function mutations affect the imd-dependent induction of antibacterial peptides and resistance to Gram-negative bacteria, whereas PGRP-LE binds to the DAP-type peptidoglycan, and a gain-of-function mutation induces constitutive activation of both the imd pathway and melanization. Here, we generated PGRP-LE null mutants and report that PGRP-LE functions synergistically with PGRP-LC in producing resistance to Escherichia coli and Bacillus megaterium infections, which have the DAP-type peptidoglycan. Consistent with this, PGRP-LE acts both upstream and in parallel with PGRP-LC in the imd pathway, and is required for infection-dependent activation of melanization in Drosophila. A role for PGRP-LE in the epithelial induction of antimicrobial peptides is also suggested.
Figures






References
-
- Akira S, Takeda K, Kaisho T (2001) Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol 2: 675–680 - PubMed
-
- Ashida M, Brey PT (1998) Recent advances in research on the insect prophenoloxidase cascade. In Molecular Mechanisms of Immune Response in Insects, Brey PT, Hultmark D (eds) pp 135–172. London: Chapman and Hall
-
- Choe KM, Werner T, Stoven S, Hultmark D, Anderson KV (2002) Requirement for a peptidoglycan recognition protein (PGRP) in Relish activation and antibacterial immune responses in Drosophila. Science 296: 359–362 - PubMed
-
- De Gregorio E, Han SJ, Lee WJ, Baek MJ, Osaki T, Kawabata S, Lee BL, Iwanaga S, Lemaitre B, Brey PT (2002a) An immune-responsive Serpin regulates the melanization cascade in Drosophila. Dev Cell 3: 581–592 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

