Comparison of enzyme-linked immunosorbent assay, Western blotting, microagglutination, indirect immunofluorescence assay, and flow cytometry for serological diagnosis of tularemia
- PMID: 15539498
- PMCID: PMC524736
- DOI: 10.1128/CDLI.11.6.1008-1015.2004
Comparison of enzyme-linked immunosorbent assay, Western blotting, microagglutination, indirect immunofluorescence assay, and flow cytometry for serological diagnosis of tularemia
Abstract
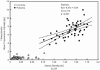
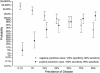
The serodiagnostic efficiencies of five different approaches to detecting antibodies (immunoglobulins G, A, and M) developed in clinically proven infections with Francisella tularensis have been assessed. Fifty serum samples from patients suffering from tularemia during an outbreak in Sweden were compared with samples from 50 healthy blood donors (controls) by using an enzyme-linked immunosorbent assay (ELISA), microagglutination (MA), Western blotting (WB), an indirect immunofluorescence assay (IIFA), and flow cytometry (FC). ELISA, WB, and FC were based on the use of preparations of lipopolysaccharides (LPS) of the live vaccine strain of Francisella tularensis subsp. holarctica (ATCC 29684) as a capture antigen. Whole methanol-fixed bacteria were used for IIFA and MA. Optimized protocols yielded a diagnostic sensitivity and specificity of 100% for WB, MA, and FC, 98% for ELISA, and 93% for IIFA. A total of 6,632 serum samples from individuals between the ages of 18 and 79 years, representatively recruited from all regions of Germany, were screened to estimate and confirm the positive predictive value (PVpos) of the ELISA. Serum samples from 15 (0.226%) individuals tested positive for F. tularensis-specific antibodies by ELISA and confirmatory WB. The resulting prevalence-dependent PVpos of 10.2% and specificity of 98.1% were consistent with our findings for tularemia patients and controls. We conclude that the combined usage of a screening ELISA and a confirmatory WB based on LPS as a common antigen, as well as the MA, is a suitable serodiagnostic tool, while the quality of the IIFA is hampered by subjective variations of the results. FC is a promising new approach that might be improved further in terms of multiplex analyses or high-throughput applications.
Figures



Similar articles
-
A novel screening ELISA and a confirmatory Western blot useful for diagnosis and epidemiological studies of tularemia.Epidemiol Infect. 2005 Aug;133(4):759-66. doi: 10.1017/s0950268805003742. Epidemiol Infect. 2005. PMID: 16050523 Free PMC article.
-
[Tularemia seroprevalence in the risky population living in both rural and urban areas of Erzurum].Mikrobiyol Bul. 2011 Jan;45(1):67-74. Mikrobiyol Bul. 2011. PMID: 21341161 Turkish.
-
Detection of Francisella tularensis-specific antibodies in patients with tularemia by a novel competitive enzyme-linked immunosorbent assay.Clin Vaccine Immunol. 2013 Jan;20(1):9-16. doi: 10.1128/CVI.00516-12. Epub 2012 Oct 31. Clin Vaccine Immunol. 2013. PMID: 23114700 Free PMC article.
-
Francisella tularensis, Tularemia and Serological Diagnosis.Front Cell Infect Microbiol. 2020 Oct 26;10:512090. doi: 10.3389/fcimb.2020.512090. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 33194778 Free PMC article. Review.
-
Diagnostic procedures in tularaemia with special focus on molecular and immunological techniques.J Vet Med B Infect Dis Vet Public Health. 2005 Aug;52(6):249-61. doi: 10.1111/j.1439-0450.2005.00863.x. J Vet Med B Infect Dis Vet Public Health. 2005. PMID: 16219088 Review.
Cited by
-
Bacillus anthracis, Francisella tularensis and Yersinia pestis. The most important bacterial warfare agents - review.Folia Microbiol (Praha). 2009;54(4):263-72. doi: 10.1007/s12223-009-0046-1. Epub 2009 Oct 14. Folia Microbiol (Praha). 2009. PMID: 19826916 Review.
-
A Review on Important Zoonotic Bacterial Tick-Borne Diseases in the Eastern Mediterranean Region.J Arthropod Borne Dis. 2021 Sep 30;15(3):265-277. doi: 10.18502/jad.v15i3.9814. eCollection 2021 Sep. J Arthropod Borne Dis. 2021. PMID: 36578998 Free PMC article. Review.
-
Arbobacteria - Pathogens Transmittable by Arthropods.Transfus Med Hemother. 2009;36(1):62-78. doi: 10.1159/000197341. Transfus Med Hemother. 2009. PMID: 21048822 Free PMC article. No abstract available.
-
Performance of seven serological assays for diagnosing tularemia.BMC Infect Dis. 2014 May 5;14:234. doi: 10.1186/1471-2334-14-234. BMC Infect Dis. 2014. PMID: 24885274 Free PMC article.
-
Francisella tularensis Subspecies holarctica and Tularemia in Germany.Microorganisms. 2020 Sep 22;8(9):1448. doi: 10.3390/microorganisms8091448. Microorganisms. 2020. PMID: 32971773 Free PMC article. Review.
References
-
- Aronova, N. V., and N. V. Pavlovich. 2001. Comparative analysis of the immune response of a rabbit to antigens to live and killed Francisella species bacteria. Mol. Gen. Mikrobiol. Virusol. 2001:26-30. (In Russian.) - PubMed
-
- Bellach, B. M., H. Knopf, and W. Thefeld. 1998. The German Health Survey, 1997/98. Gesundheitswesen 60(Suppl. 2):S59-S68. (In German.) - PubMed
-
- Berdal, B. P., R. Mehl, H. Haaheim, M. Loksa, R. Grunow, J. Burans, C. Morgan, and H. Meyer. 2000. Field detection of Francisella tularensis. Scand. J. Infect. Dis. 32:287-291. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

