Cytokines--central factors in alcoholic liver disease
- PMID: 15540802
- PMCID: PMC6668870
Cytokines--central factors in alcoholic liver disease
Abstract
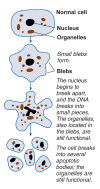
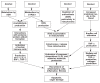
Many processes related to the consumption or breakdown of alcohol that contribute to alcohol-induced liver disease are mediated by small proteins known as cytokines, which are produced and secreted by liver cells and many other cells throughout the body. Through a variety of actions, cytokines regulate certain biochemical. processes in the cells that produce them as well as in neighboring cells. For example, in case of an infection, they attract white blood cells to the tissues, triggering an inflammatory response. In the liver, persistent cytokine secretion resulting in chronic inflammation leads to conditions such as hepatitis, fibrosis, and cirrhosis. Cytokines also regulate a process known as programmed cell death, or apoptosis, which is in part responsible for alcohol-induced destruction of liver tissue. Two cytokines-tumor necrosis factor alpha and transforming growth factor beta-play prominent roles in apoptosis. Finally, a cytokine network mediates the harmful effects of a bacterial protein called endotoxin on the liver. Because of their diverse functions, cytokines might make attractive targets in the prevention or treatment of alcoholic liver disease, and researchers already have obtained encouraging results when testing such approaches.
Figures
References
-
- Bedossa P, Paradis V. Transforming growth factor-beta (TGF-beta): A key role in liver fibrogenesis. Journal of Hepatology. 1995;22(Suppl 2):37–42. - PubMed
-
- Cameron RG, Neuman MG. Novel morphologic findings in alcoholic liver disease. Clinical Biochemistry. 1999;32:579–584. - PubMed
-
- Fernandez-Checa JC, Yi JR, Garcia-Ruiz C, et al. Plasma membrane and mitochondrial transport of hepatic reduced glutathione. Seminars in Liver Disease. 1996;16:147–158. - PubMed
-
- Garcia-Ruiz C, Colell A, Morales A, et al. Role of oxidative stress generated from the mitochondrial electron transport chain and mitochondrial glutathione status in loss of mitochondrial function and activation of transcription factor nuclear factor-kappa B: Studies with isolated mitochondria and rat hepatocytes. Molecular Pharmacology. 1995a;48(5):825–834. - PubMed
-
- Garcia-Ruiz C, Morales A, Colell A, et al. Feeding S-adenosyl-L-methionine attenuates both ethanol-induced depletion of mitochondrial glutathione and mitochondrial dysfunction in periportal and perivenous rat hepatocytes. Hepatology. 1995b;21:207–214. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
