TATA box binding protein induces structure in the recombinant glucocorticoid receptor AF1 domain
- PMID: 15545613
- PMCID: PMC534534
- DOI: 10.1073/pnas.0407160101
TATA box binding protein induces structure in the recombinant glucocorticoid receptor AF1 domain
Abstract
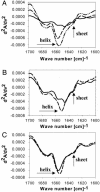
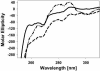
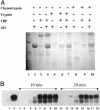
A number of transcription factor proteins contain domains that are fully or partially unstructured. The means by which such proteins acquire naturally folded conformations are not well understood. When they encounter their proper binding partner(s), several of these proteins adopt a folded conformation through an induced-fit mechanism. The glucocorticoid receptor (GR) is a ligand-activated transcription factor. Expressed independently as a recombinant peptide, the N-terminal transactivation domain (AF1) of the GR shows little structure and appears to exist as a collection of random coil configurations. The GR AF1 is known to interact with other transcription factors, including a critical component of the general transcription machinery proteins, the TATA box binding protein (TBP). We tested whether this interaction can lead to acquisition of structure in the GR AF1. Our results show that recombinant GR AF1 acquires a significant amount of helical content when it interacts with TBP. These structural changes were monitored by Fourier transform infrared and NMR spectroscopies, and by proteolytic digestions. Our results support a model in which TBP binding interaction with the GR AF1 induces significantly greater helical structure in the AF1 domain. This increased helical content in the GR AF1 appears to come mostly at the expense of random coil conformation. These results are in accordance with the hypothesis that an induced-fit mechanism gives structure to the GR AF1 when it encounters TBP.
Figures


References
-
- Beato, M. (1989) Cell 56, 335–344. - PubMed
-
- Yamamoto, K. R. (1985) Annu. Rev. Genet. 19, 209–252. - PubMed
-
- Simons, S. S., Jr. (1994) in Vitamins and Hormones, ed. Litwack, G. (Academic, London), pp. 49–130. - PubMed
-
- Freedman, L. P., Luisi, B. F., Korszun, Z. R., Basavappa, R., Sigler, P. B. & Yamamoto, K. R. (1988) Nature 334, 543–546. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

