Combinatorial antibody libraries from cancer patients yield ligand-mimetic Arg-Gly-Asp-containing immunoglobulins that inhibit breast cancer metastasis
- PMID: 15563590
- PMCID: PMC534417
- DOI: 10.1073/pnas.0407869101
Combinatorial antibody libraries from cancer patients yield ligand-mimetic Arg-Gly-Asp-containing immunoglobulins that inhibit breast cancer metastasis
Abstract
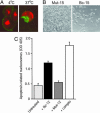
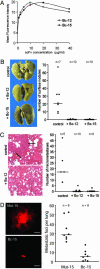
Combinatorial antibody libraries have the potential to display the entire immunological record of an individual, allowing one to detect and recover any antibody ever made, irrespective of whether it is currently being produced. We have termed this the "fossil record" of an individual's antibody response. To determine whether cancer patients have ever made antibodies with disease-fighting potential, we screened combinatorial antibody libraries from cancer patients for immunoglobulins that can identify metastatic tumor cells. This strategy yielded human antibodies specific for the activated conformation of the adhesion receptor integrin alphavbeta3 that is associated with a metastatic phenotype. In a remarkable example of convergent evolution, two of these antibodies were shown to contain the Arg-Gly-Asp integrin recognition motif of the natural ligand within the third complementarity-determining region of the heavy chain. These antibodies interfered with lung colonization by human breast cancer cells in a mouse model and inhibited existing metastatic disease. Our data imply that, at least at some time, these antibodies were part of a patient's surveillance system against metastatic cells, targeting the activated conformer of integrin alphavbeta3 and disrupting its functions. The ligand-mimetic nature of these antibodies, combined with specificity for a single receptor, is unique in the integrin-ligand repertoire. The convergent evolution of critical sequences in antibodies and other ligands that bind to the same target means that the immune response has sufficient power to find a best chemical solution for the optimization of binding energy, even though antibodies evolve in real time, as compared with billions of years for the natural ligand.
Figures
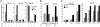


Similar articles
-
Targeting activated integrin alphavbeta3 with patient-derived antibodies impacts late-stage multiorgan metastasis.Clin Exp Metastasis. 2010 Apr;27(4):217-31. doi: 10.1007/s10585-010-9320-5. Epub 2010 Mar 12. Clin Exp Metastasis. 2010. PMID: 20225083 Free PMC article.
-
Selective interaction of a conformationally-constrained Arg-Gly-Asp (RGD) motif with the integrin receptor alphavbeta3 expressed on human tumor cells.Blood Cells Mol Dis. 1997 Aug;23(2):230-41. doi: 10.1006/bcmd.1997.0140. Blood Cells Mol Dis. 1997. PMID: 9268674
-
A peptide mimic of E-selectin ligand inhibits sialyl Lewis X-dependent lung colonization of tumor cells.Cancer Res. 2000 Jan 15;60(2):450-6. Cancer Res. 2000. PMID: 10667600
-
From combinatorial chemistry to cancer-targeting peptides.Mol Pharm. 2007 Sep-Oct;4(5):631-51. doi: 10.1021/mp700073y. Epub 2007 Sep 20. Mol Pharm. 2007. PMID: 17880166 Review.
-
Role of platelets, thrombin, integrin IIb-IIIa, fibronectin and von Willebrand factor on tumor adhesion in vitro and metastasis in vivo.Thromb Haemost. 1995 Jul;74(1):282-90. Thromb Haemost. 1995. PMID: 8578473 Review.
Cited by
-
Efficient Exploration of Membrane-Associated Phenomena at Atomic Resolution.J Membr Biol. 2015 Jun;248(3):563-82. doi: 10.1007/s00232-015-9806-9. Epub 2015 May 22. J Membr Biol. 2015. PMID: 25998378 Free PMC article. Review.
-
Activated tumor cell integrin αvβ3 cooperates with platelets to promote extravasation and metastasis from the blood stream.Thromb Res. 2016 Apr;140 Suppl 1(Suppl 1):S27-36. doi: 10.1016/S0049-3848(16)30095-0. Thromb Res. 2016. PMID: 27067975 Free PMC article.
-
Breaking the one antibody-one target axiom.Proc Natl Acad Sci U S A. 2006 Jul 18;103(29):11009-14. doi: 10.1073/pnas.0603822103. Epub 2006 Jul 5. Proc Natl Acad Sci U S A. 2006. PMID: 16822849 Free PMC article.
-
Targeting activated integrin alphavbeta3 with patient-derived antibodies impacts late-stage multiorgan metastasis.Clin Exp Metastasis. 2010 Apr;27(4):217-31. doi: 10.1007/s10585-010-9320-5. Epub 2010 Mar 12. Clin Exp Metastasis. 2010. PMID: 20225083 Free PMC article.
-
Healthy humans can be a source of antibodies countering COVID-19.Bioengineered. 2022 May;13(5):12598-12624. doi: 10.1080/21655979.2022.2076390. Bioengineered. 2022. PMID: 35599623 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

