Liver-targeted disruption of Apc in mice activates beta-catenin signaling and leads to hepatocellular carcinomas
- PMID: 15563600
- PMCID: PMC535370
- DOI: 10.1073/pnas.0404761101
Liver-targeted disruption of Apc in mice activates beta-catenin signaling and leads to hepatocellular carcinomas
Abstract
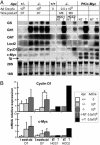
Although inappropriate activation of the Wnt/beta-catenin pathway has been implicated in the development of hepatocellular carcinoma (HCC), the role of this signaling in liver carcinogenesis remains unclear. To investigate this issue, we constructed a mutant mouse strain, Apc(lox/lox), in which exon 14 of the tumor-suppressor gene adenomatous polyposis coli (Apc) is flanked by loxP sequences. i.v. injection of adenovirus encoding Cre recombinase (AdCre) at high multiplicity [10(9) plaque-forming units (pfu) per mouse] inactivated the Apc gene in the liver and resulted in marked hepatomegaly, hepatocyte hyperplasia, and rapid mortality. beta-Catenin signaling activation was demonstrated by nuclear and cytoplasmic accumulation of beta-catenin in the hepatocytes and by the induction of beta-catenin target genes (glutamine synthetase, glutamate transporter 1, ornithine aminotransferase, and leukocyte cell-derived chemotaxin 2) in the liver. To test a long-term oncogenic effect, we inoculated mice with lower doses of AdCre (0.5 x 10(9) pfu per mouse), compatible with both survival and persistence of beta-catenin-activated cells. In these conditions, 67% of mice developed HCC. beta-Catenin signaling was strongly activated in these Apc-inactivated HCCs. The HCCs were well, moderately, or poorly differentiated. Indeed, their histological and molecular features mimicked human HCC. Thus, deletion of Apc in the liver provides a valuable model of human HCC, and, in this model, activation of the Wnt/beta-catenin pathway by invalidation of Apc is required for liver tumorigenesis.
Figures




References
-
- Befeler, A. S. & Di Bisceglie, A. M. (2002) Gastroenterology 122, 1609–1619. - PubMed
-
- Buendia, M. A. (2000) Semin. Cancer Biol. 10, 185–200. - PubMed
-
- Giles, R. H., van Es, J. H. & Clevers, H. (2003) Biochim. Biophys. Acta 1653, 1–24. - PubMed
-
- Chen, T. C., Hsieh, L. L., Ng, K. F., Jeng, L. B. & Chen, M. F. (1998) Cancer Lett. 134, 23–28. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

