Semenogelins I and II bind zinc and regulate the activity of prostate-specific antigen
- PMID: 15563730
- PMCID: PMC1134973
- DOI: 10.1042/BJ20041424
Semenogelins I and II bind zinc and regulate the activity of prostate-specific antigen
Abstract
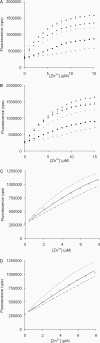
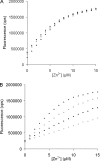
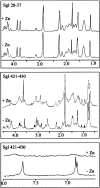
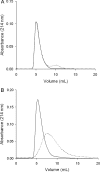
In semen, the gel proteins SgI and SgII (semenogelins I and II) are digested by PSA (prostate-specific antigen), resulting in liquefaction and release of motile spermatozoa. Semen contains a high concentration of Zn2+, which is known to inhibit the protease activity of PSA. We characterized the binding of Zn2+ to SgI and SgII and found evidence that these proteins are involved in regulating the activity of PSA. Intact SgI and SgII and synthetic semenogelin peptides were used in the experiments. Binding of Zn2+ was studied by radioligand blotting, titration with a zinc (II) fluorophore chelator and NMR analysis. A chromogenic substrate was used to measure the enzymatic activity of PSA. SgI and SgII bound Zn2+ with a stoichiometry of at least 10 mol (mol of protein)(-1) and with an average dissociation constant of approx. 5 microM per site. Moreover, Zn2+-inhibited PSA was activated by exposure to SgI or SgII. Since both proteins have high affinity for Zn2+ and are the dominating proteins in semen, they probably represent the major Zn2+ binders in semen, one function of which may be to regulate the activity of PSA. The system is self-regulating, and PSA is maintained in an active state by its substrate.
Figures

Similar articles
-
Characterization of prostate-specific antigen proteolytic activity on its major physiological substrate, the sperm motility inhibitor precursor/semenogelin I.Biochemistry. 1997 Apr 1;36(13):3811-9. doi: 10.1021/bi9626158. Biochemistry. 1997. PMID: 9092810
-
Truncated semenogelin I binds zinc and is cleaved by prostate-specific antigen.J Androl. 2006 Jul-Aug;27(4):542-7. doi: 10.2164/jandrol.05188. Epub 2006 Apr 1. J Androl. 2006. PMID: 16582407
-
Ejaculates from the common marmoset (Callithrix jacchus) contain semenogelin and beta-microseminoprotein but not prostate-specific antigen.Mol Reprod Dev. 2005 Jun;71(2):247-55. doi: 10.1002/mrd.20257. Mol Reprod Dev. 2005. PMID: 15791587
-
The interaction among protein C inhibitor, prostate-specific antigen, and the semenogelin system.Semin Thromb Hemost. 2007 Feb;33(1):46-52. doi: 10.1055/s-2006-958461. Semin Thromb Hemost. 2007. PMID: 17253189 Review.
-
Semenogelin, the main protein of the human semen coagulum, regulates sperm function.Semin Thromb Hemost. 2007 Feb;33(1):60-8. doi: 10.1055/s-2006-958463. Semin Thromb Hemost. 2007. PMID: 17253191 Review.
Cited by
-
The major bactericidal activity of human seminal plasma is zinc-dependent and derived from fragmentation of the semenogelins.J Immunol. 2008 Sep 1;181(5):3413-21. doi: 10.4049/jimmunol.181.5.3413. J Immunol. 2008. PMID: 18714013 Free PMC article.
-
Semenogelins in the human retina: Differences in distribution and content between AMD and normal donor tissues.Exp Eye Res. 2008 Jan;86(1):150-6. doi: 10.1016/j.exer.2007.10.006. Epub 2007 Oct 25. Exp Eye Res. 2008. PMID: 18036592 Free PMC article.
-
Correlation of epididymal protease inhibitor and fibronectin in human semen.PLoS One. 2013 Dec 16;8(12):e82600. doi: 10.1371/journal.pone.0082600. eCollection 2013. PLoS One. 2013. PMID: 24358212 Free PMC article.
-
Mutations in the prostate specific antigen (PSA/KLK3) correlate with male infertility.Sci Rep. 2017 Sep 11;7(1):11225. doi: 10.1038/s41598-017-10866-1. Sci Rep. 2017. PMID: 28894123 Free PMC article.
-
Proteomic changes of alveolar lining fluid in illnesses associated with exposure to inhaled non-infectious microbial particles.PLoS One. 2014 Jul 17;9(7):e102624. doi: 10.1371/journal.pone.0102624. eCollection 2014. PLoS One. 2014. PMID: 25033447 Free PMC article.
References
-
- Lilja H., Abrahamsson P. A., Lundwall A. Semenogelin, the predominant protein in human semen. Primary structure and identification of closely related proteins in the male accessory sex glands and on the spermatozoa. J. Biol. Chem. 1989;264:1894–1900. - PubMed
-
- Robert M., Gibbs B. F., Jacobson E., Gagnon C. Characterization of prostate-specific antigen proteolytic activity on its major physiological substrate, the sperm motility inhibitor precursor/semenogelin I. Biochemistry. 1997;36:3811–3819. - PubMed
-
- Malm J., Hellman J., Magnusson H., Laurell C. B., Lilja H. Isolation and characterization of the major gel proteins in human semen, semenogelin I and semenogelin II. Eur. J. Biochem. 1996;238:48–53. - PubMed
-
- Ulvsback M., Lazure C., Lilja H., Spurr N. K., Rao V. V., Loffler C., Hansmann I., Lundwall A. Gene structure of semenogelin I and II. The predominant proteins in human semen are encoded by two homologous genes on chromosome 20. J. Biol. Chem. 1992;267:18080–18084. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

