Calcium-independent calcium/calmodulin-dependent protein kinase II in the adult Drosophila CNS enhances the training of pheromonal cues
- PMID: 15564574
- PMCID: PMC6730130
- DOI: 10.1523/JNEUROSCI.3560-04.2004
Calcium-independent calcium/calmodulin-dependent protein kinase II in the adult Drosophila CNS enhances the training of pheromonal cues
Abstract
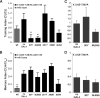
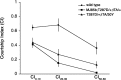
Calcium/calmodulin-dependent protein kinase II (CaMKII) is abundant in the CNS and is crucial for cellular and behavioral plasticity. It is thought that the ability of CaMKII to autophosphorylate and become Ca2+ independent allows it to act as a molecular memory switch. We have shown previously that inhibition of Drosophila CaMKII leads to impaired performance in the courtship conditioning associative memory assay, but it was unknown whether the constitutive form of the kinase had a special role in learning. In this study, we use a tripartite transgenic system combining GAL4/UAS with the tetracycline-off system to spatially and temporally manipulate levels of Ca2+-independent CaMKII activity in Drosophila. We find an enhancement of information processing during the training period with Ca2+-independent, but not Ca2+-dependent, CaMKII. During training, control animals have a lag before active suppression of courtship begins. Animals expressing Ca2+-independent CaMKII have no lag, implying that there is a threshold level of Ca2+-independent activity that must be present to suppress courtship. This is the first demonstration, in any organism, of enhanced behavioral plasticity with overexpression of constitutively active CaMKII. Anatomical studies indicate that transgene expression in antennal lobes and extrinsic mushroom body neurons drives this behavioral enhancement. Interestingly, immediate memory was unaffected by expression of T287D CaMKII in mushroom bodies, although previous studies have shown that CaMKII activity is required in this brain region for memory formation. These results suggest that the biochemical mechanisms of CaMKII-dependent memory formation are threshold based in only a subset of neurons.
Figures




Similar articles
-
Cholinergic neurons mediate CaMKII-dependent enhancement of courtship suppression.Learn Mem. 2006 Nov-Dec;13(6):686-9. doi: 10.1101/lm.317806. Epub 2006 Nov 13. Learn Mem. 2006. PMID: 17101876
-
Visual input regulates circuit configuration in courtship conditioning of Drosophila melanogaster.Learn Mem. 2000 Jan;7(1):32-42. doi: 10.1101/lm.7.1.32. Learn Mem. 2000. PMID: 10706600 Free PMC article.
-
Mapping of the anatomical circuit of CaM kinase-dependent courtship conditioning in Drosophila.Learn Mem. 1999 Mar-Apr;6(2):177-92. Learn Mem. 1999. PMID: 10327242 Free PMC article.
-
A fresh look at the role of CaMKII in hippocampal synaptic plasticity and memory.Bioessays. 2002 Mar;24(3):223-33. doi: 10.1002/bies.10057. Bioessays. 2002. PMID: 11891759 Review.
-
Calcium/calmodulin-dependent protein kinase II and synaptic plasticity.Curr Opin Neurobiol. 2004 Jun;14(3):318-27. doi: 10.1016/j.conb.2004.05.008. Curr Opin Neurobiol. 2004. PMID: 15194112 Review.
Cited by
-
CASK and CaMKII function in Drosophila memory.Front Neurosci. 2014 Jun 25;8:178. doi: 10.3389/fnins.2014.00178. eCollection 2014. Front Neurosci. 2014. PMID: 25009461 Free PMC article. Review.
-
Constitutive activation of Ca2+/calmodulin-dependent protein kinase II during development impairs central cholinergic transmission in a circuit underlying escape behavior in Drosophila.J Neurosci. 2012 Jan 4;32(1):170-82. doi: 10.1523/JNEUROSCI.6583-10.2012. J Neurosci. 2012. PMID: 22219280 Free PMC article.
-
The histone deacetylase HDAC4 regulates long-term memory in Drosophila.PLoS One. 2013 Dec 9;8(12):e83903. doi: 10.1371/journal.pone.0083903. eCollection 2013. PLoS One. 2013. PMID: 24349558 Free PMC article.
-
Orai-mediated calcium entry determines activity of central dopaminergic neurons by regulation of gene expression.Elife. 2024 Jan 30;12:RP88808. doi: 10.7554/eLife.88808. Elife. 2024. PMID: 38289659 Free PMC article.
-
Mating alters gene expression patterns in Drosophila melanogaster male heads.BMC Genomics. 2010 Oct 11;11:558. doi: 10.1186/1471-2164-11-558. BMC Genomics. 2010. PMID: 20937114 Free PMC article.
References
-
- Bach ME, Hawkins RD, Osman M, Kandel ER, Mayford M (1995) Impairment of spatial but not contextual memory in CaMKII mutant mice with a selective loss of hippocampal LTP in the range of the theta frequency. Cell 81: 905-915. - PubMed
-
- Bello B, Resendez-Perez D, Gehring WJ (1998) Spatial and temporal targeting of gene expression in Drosophila by means of a tetracycline-dependent transactivator system. Development 125: 2193-2202. - PubMed
-
- Brand AH, Dormand EL (1995) The GAL4 system as a tool for unraveling the mysteries of the Drosophila nervous system. Curr Opin Neurobiol 5: 572-578. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
