Immunodominant PstS1 antigen of mycobacterium tuberculosis is a potent biological response modifier for the treatment of bladder cancer
- PMID: 15566565
- PMCID: PMC544192
- DOI: 10.1186/1471-2407-4-86
Immunodominant PstS1 antigen of mycobacterium tuberculosis is a potent biological response modifier for the treatment of bladder cancer
Abstract
Background: Bacillus Calmette Guerin (BCG)-immunotherapy has a well-documented and successful clinical history in the treatment of bladder cancer. However, regularly observed side effects, a certain degree of nonresponders and restriction to superficial cancers remain a major obstacle. Therefore, alternative treatment strategies are intensively being explored. We report a novel approach of using a well defined immunostimulatory component of Mycobacterium tuberculosis for the treatment of bladder cancer. The phosphate transport protein PstS1 which represents the phosphate binding component of a mycobacterial phosphate uptake system is known to be a potent immunostimulatory antigen of M. tuberculosis. This preclinical study was designed to test the potential of recombinant PstS1 to serve as a non-viable and defined immunotherapeutic agent for intravesical bladder cancer therapy.
Methods: Mononuclear cells (PBMCs) were isolated from human peripheral blood and stimulated with PstS1 for seven days. The activation of PBMCs was determined by chromium release assay, IFN-gamma ELISA and measurement of lymphocyte proliferation. The potential of PstS1 to activate monocyte-derived human dendritic cells (DC) was determined by flow cytometric analysis of the marker molecules CD83 and CD86 as well as the release of the cytokines TNF-alpha and IL-12. Survival of presensitized and intravesically treated, tumor-bearing mice was analyzed by Kaplan-Meier curve and log rank test. Local and systemic immune response in PstS1-immunotherapy was investigated by anti-PstS1-specific ELISA, splenocyte proliferation assay and immunohistochemistry.
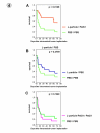
Results: Our in vitro experiments showed that PstS1 is able to stimulate cytotoxicity, IFN-gamma release and proliferation of PBMCs. Further investigations showed the potential of PstS1 to activate monocyte-derived human dendritic cells (DC). In vivo studies in an orthotopic murine bladder cancer model demonstrated the therapeutic potential of intravesically applied PstS1. Immunohistochemical analysis and splenocyte restimulation assay revealed that local and systemic immune responses were triggered by intravesical PstS1-immunotherapy.
Conclusion: Our results demonstrate profound in vitro activation of human immune cells by recombinant PstS1. In addition, intravesical PstS1 immunotherapy induced strong local and systemic immune responses together with substantial anti-tumor activity in a preclinical mouse model. Thus, we have identified recombinant PstS1 antigen as a potent immunotherapeutic drug for cancer therapy.
Figures





References
-
- Lamm DL, Blumenstein BA, Crawford ED, Montie JE, Scardino P, Grossman HB, Stanisic TH, Smith JAJ, Sullivan J, Sarosdy MF, . A randomized trial of intravesical doxorubicin and immunotherapy with bacille Calmette-Guerin for transitional-cell carcinoma of the bladder. N Engl J Med. 1991;325:1205–1209. - PubMed
-
- Herr HW, Schwalb DM, Zhang ZF, Sogani PC, Fair WR, Whitmore WFJ, Oettgen HF. Intravesical bacillus Calmette-Guerin therapy prevents tumor progression and death from superficial bladder cancer: ten-year follow-up of a prospective randomized trial. J Clin Oncol. 1995;13:1404–1408. - PubMed
-
- Malmstrom PU, Wijkstrom H, Lundholm C, Wester K, Busch C, Norlen BJ. 5-year followup of a randomized prospective study comparing mitomycin C and bacillus Calmette-Guerin in patients with superficial bladder carcinoma. Swedish-Norwegian Bladder Cancer Study Group. J Urol. 1999;161:1124–1127. doi: 10.1097/00005392-199904000-00019. - DOI - PubMed
-
- Herr HW, Badalament RA, Amato DA, Laudone VP, Fair WR, Whitmore WFJ. Superficial bladder cancer treated with bacillus Calmette-Guerin: a multivariate analysis of factors affecting tumor progression. J Urol. 1989;141:22–29. - PubMed
-
- Cymes M, Fleischmann JD, Smith E. Invasive bladder cancer treated with intravesical BCG. J Urol. 1992;147:273.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

