HIV-1 Nef disrupts MHC-I trafficking by recruiting AP-1 to the MHC-I cytoplasmic tail
- PMID: 15569716
- PMCID: PMC2172469
- DOI: 10.1083/jcb.200407031
HIV-1 Nef disrupts MHC-I trafficking by recruiting AP-1 to the MHC-I cytoplasmic tail
Abstract
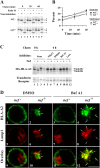
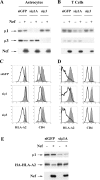
To avoid immune recognition by cytotoxic T lymphocytes (CTLs), human immunodeficiency virus (HIV)-1 Nef disrupts the transport of major histocompatibility complex class I molecules (MHC-I) to the cell surface in HIV-infected T cells. However, the mechanism by which Nef does this is unknown. We report that Nef disrupts MHC-I trafficking by rerouting newly synthesized MHC-I from the trans-Golgi network (TGN) to lysosomal compartments for degradation. The ability of Nef to target MHC-I from the TGN to lysosomes is dependent on expression of the mu1 subunit of adaptor protein (AP) AP-1A, a cellular protein complex implicated in TGN to endolysosomal pathways. We demonstrate that in HIV-infected primary T cells, Nef promotes a physical interaction between endogenous AP-1 and MHC-I. Moreover, we present data that this interaction uses a novel AP-1 binding site that requires amino acids in the MHC-I cytoplasmic tail. In sum, our evidence suggests that binding of AP-1 to the Nef-MHC-I complex is an important step required for inhibition of antigen presentation by HIV.
Figures

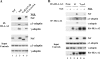



References
-
- Aiken, C., J. Konner, N.R. Landau, M.E. Lenburg, and D. Trono. 1994. Nef induces CD4 endocytosis: requirement for a critical dileucine motif in the membrane-proximal CD4 cytoplasmic domain. Cell. 76:853–864. - PubMed
-
- Alexander, M., Y.C. Bor, K.S. Ravichandran, M.L. Hammarskjold, and D. Rekosh. 2004. Human immunodeficiency virus type 1 Nef associates with lipid rafts to downmodulate cell surface CD4 and class I major histocompatibility complex expression and to increase viral infectivity. J. Virol. 78:1685–1696. - PMC - PubMed
-
- Blagoveshchenskaya, A.D., L. Thomas, S.F. Feliciangeli, C.H. Hung, and G. Thomas. 2002. HIV-1 Nef downregulates MHC-I by a PACS-1- and PI3K-regulated ARF6 endocytic pathway. Cell. 111:853–866. - PubMed
-
- Bonifacino, J.S., and L.M. Traub. 2003. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 72:395–447. - PubMed
-
- Bresnahan, P.A., W. Yonemoto, S. Ferrell, D. Williams-Herman, R. Geleziunas, and W.C. Greene. 1998. A dileucine motif in HIV-1 Nef acts as an internalization signal for CD4 downregulation and binds the AP-1 clathrin adaptor. Curr. Biol. 8:1235–1238. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

