Laminin-5 induces osteogenic gene expression in human mesenchymal stem cells through an ERK-dependent pathway
- PMID: 15574877
- PMCID: PMC545919
- DOI: 10.1091/mbc.e04-08-0695
Laminin-5 induces osteogenic gene expression in human mesenchymal stem cells through an ERK-dependent pathway
Abstract
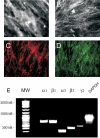
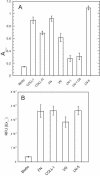
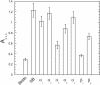
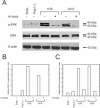
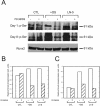
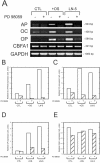
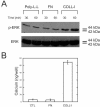
The laminin family of proteins is critical for managing a variety of cellular activities including migration, adhesion, and differentiation. In bone, the roles of laminins in controlling osteogenic differentiation of human mesenchymal stem cells (hMSC) are unknown. We report here that laminin-5 is found in bone and expressed by hMSC. hMSC isolated from bone synthesize laminin-5 and adhere to exogenous laminin-5 through alpha3beta1 integrin. Adhesion to laminin-5 activates extracellular signal-related kinase (ERK) within 30 min and leads to phosphorylation of the osteogenic transcription factor Runx2/CBFA-1 within 8 d. Cells plated on laminin-5 for 16 d express increased levels of osteogenic marker genes, and those plated for 21 d deposit a mineralized matrix, indicative of osteogenic differentiation. Addition of the ERK inhibitor PD98059 mitigates these effects. We conclude that contact with laminin-5 is sufficient to activate ERK and to stimulate osteogenic differentiation in hMSC.
Figures


Similar articles
-
Activation of FAK is necessary for the osteogenic differentiation of human mesenchymal stem cells on laminin-5.J Cell Biochem. 2007 Feb 1;100(2):499-514. doi: 10.1002/jcb.21074. J Cell Biochem. 2007. PMID: 16927379
-
Focal adhesion kinase signaling pathways regulate the osteogenic differentiation of human mesenchymal stem cells.Exp Cell Res. 2007 Jan 1;313(1):22-37. doi: 10.1016/j.yexcr.2006.09.013. Epub 2006 Sep 22. Exp Cell Res. 2007. PMID: 17081517 Free PMC article.
-
Dissection of the osteogenic effects of laminin-332 utilizing specific LG domains: LG3 induces osteogenic differentiation, but not mineralization.Exp Cell Res. 2008 Feb 15;314(4):763-73. doi: 10.1016/j.yexcr.2007.12.007. Epub 2008 Jan 22. Exp Cell Res. 2008. PMID: 18206871 Free PMC article.
-
ERK signaling pathways regulate the osteogenic differentiation of human mesenchymal stem cells on collagen I and vitronectin.Cell Commun Adhes. 2004 Sep-Dec;11(5-6):137-53. doi: 10.1080/15419060500242836. Cell Commun Adhes. 2004. PMID: 16194881
-
Laminins in osteogenic differentiation and pluripotency maintenance.Differentiation. 2020 Jul-Aug;114:13-19. doi: 10.1016/j.diff.2020.05.002. Epub 2020 May 16. Differentiation. 2020. PMID: 32473527 Review.
Cited by
-
Mesenchymal Stromal Cells as Critical Contributors to Tissue Regeneration.Front Cell Dev Biol. 2020 Sep 25;8:576176. doi: 10.3389/fcell.2020.576176. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33102483 Free PMC article. Review.
-
Tailored integrin-extracellular matrix interactions to direct human mesenchymal stem cell differentiation.Stem Cells Dev. 2012 Sep 1;21(13):2442-56. doi: 10.1089/scd.2011.0615. Epub 2012 May 31. Stem Cells Dev. 2012. PMID: 22455378 Free PMC article.
-
How Mechanical and Physicochemical Material Characteristics Influence Adipose-Derived Stem Cell Fate.Int J Mol Sci. 2023 Feb 10;24(4):3551. doi: 10.3390/ijms24043551. Int J Mol Sci. 2023. PMID: 36834966 Free PMC article. Review.
-
Strategies to retain properties of bone marrow-derived mesenchymal stem cells ex vivo.Ann N Y Acad Sci. 2017 Dec;1409(1):3-17. doi: 10.1111/nyas.13451. Epub 2017 Oct 6. Ann N Y Acad Sci. 2017. PMID: 28984359 Free PMC article. Review.
-
Epitope mapping of function-blocking monoclonal antibody CM6 suggests a "weak" integrin binding site on the laminin-332 LG2 domain.J Cell Physiol. 2010 Jun;223(3):541-8. doi: 10.1002/jcp.22107. J Cell Physiol. 2010. PMID: 20301201 Free PMC article.
References
-
- Aberdam, D., Aguzzi, A., Baudoin, C., Galliano, M. F., Ortonne, J. P., and Meneguzzi, G. (1994). Developmental expression of nicein adhesion protein (laminin-5) subunits suggests multiple morphogenic roles. Cell Adhes. Commun. 2, 115-129. - PubMed
-
- Aubin, J. E. (1998). Advances in the osteoblast lineage. Biochem. Cell Biol. 76, 899-910. - PubMed
-
- Barry, F. P., and Murphy, J. M. (2004). Mesenchymal stem cells: clinical applications and biological characterization. Int. J. Biochem. Cell Biol. 36, 568-584. - PubMed
-
- Belkin, A. M., and Stepp, M. A. (2000). Integrins as receptors for laminins. Microsc. Res. Technol. 51, 280-301. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

