In vivo bypass of chaperone by extended coiled-coil motif in T4 tail fiber
- PMID: 15576786
- PMCID: PMC532435
- DOI: 10.1128/JB.186.24.8363-8369.2004
In vivo bypass of chaperone by extended coiled-coil motif in T4 tail fiber
Abstract
The distal-half tail fiber of bacteriophage T4 is made of three gene products: trimeric gp36 and gp37 and monomeric gp35. Chaperone P38 is normally required for folding gp37 peptides into a P37 trimer; however, a temperature-sensitive mutation in T4 (ts3813) that suppresses this requirement at 30 degrees C but not at 42 degrees C was found in gene 37 (R. J. Bishop and W. B. Wood, Virology 72:244-254, 1976). Sequencing of the temperature-sensitive mutant revealed a 21-bp duplication of wild-type gene 37 inserted into its C-terminal portion (S. Hashemolhosseini et al., J. Mol. Biol. 241:524-533, 1994). We noticed that the 21-amino-acid segment encompassing this duplication in the ts3813 mutant has a sequence typical of a coiled coil and hypothesized that its extension would relieve the temperature sensitivity of the ts3813 mutation. To test our hypothesis, we crossed the T4 ts3813 mutant with a plasmid encoding an engineered pentaheptad coiled coil. Each of the six mutants that we examined retained two amber mutations in gene 38 and had a different coiled-coil sequence varying from three to five heptads. While the sequences varied, all maintained the heptad-repeating coiled-coil motif and produced plaques at up to 50 degrees C. This finding strongly suggests that the coiled-coil motif is a critical factor in the folding of gp37. The presence of a terminal coiled-coil-like sequence in the tail fiber genes of 17 additional T-even phages implies the conservation of this mechanism. The increased melting temperature should be useful for "clamps" to initiate the folding of trimeric beta-helices in vitro and as an in vivo screen to identify, sequence, and characterize trimeric coiled coils.
Figures
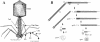

Similar articles
-
Bacteriophage T4 long tail fiber domains.Biophys Rev. 2018 Apr;10(2):463-471. doi: 10.1007/s12551-017-0348-5. Epub 2017 Dec 4. Biophys Rev. 2018. PMID: 29204885 Free PMC article. Review.
-
Two-chaperone assisted soluble expression and purification of the bacteriophage T4 long tail fibre protein gp37.Protein Expr Purif. 2010 Mar;70(1):116-21. doi: 10.1016/j.pep.2009.11.005. Epub 2009 Nov 12. Protein Expr Purif. 2010. PMID: 19913618
-
Stoichiometry and domainal organization of the long tail-fiber of bacteriophage T4: a hinged viral adhesin.J Mol Biol. 1996 Aug 2;260(5):767-80. doi: 10.1006/jmbi.1996.0436. J Mol Biol. 1996. PMID: 8709154
-
Structure of bacteriophage T4 gene product 11, the interface between the baseplate and short tail fibers.J Mol Biol. 2000 Aug 25;301(4):975-85. doi: 10.1006/jmbi.2000.3989. J Mol Biol. 2000. PMID: 10966799
-
Review: conformation and folding of novel beta-structural elements in viral fiber proteins: the triple beta-spiral and triple beta-helix.J Struct Biol. 2002 Jan-Feb;137(1-2):236-47. doi: 10.1006/jsbi.2002.4447. J Struct Biol. 2002. PMID: 12064949 Review.
Cited by
-
Characterization of extended-host-range pseudo-T-even bacteriophage Kpp95 isolated on Klebsiella pneumoniae.Appl Environ Microbiol. 2007 Apr;73(8):2532-40. doi: 10.1128/AEM.02113-06. Epub 2007 Mar 2. Appl Environ Microbiol. 2007. PMID: 17337566 Free PMC article.
-
vB_BcM_Sam46 and vB_BcM_Sam112, members of a new bacteriophage genus with unusual small terminase structure.Sci Rep. 2021 Jun 9;11(1):12173. doi: 10.1038/s41598-021-91289-x. Sci Rep. 2021. PMID: 34108535 Free PMC article.
-
Lean forward: Genetic analysis of temperature-sensitive mutants unfolds the secrets of oligomeric protein complex assembly.Bioessays. 2014 Sep;36(9):836-46. doi: 10.1002/bies.201400062. Epub 2014 Jul 22. Bioessays. 2014. PMID: 25048147 Free PMC article.
-
Bacteriophage T4 long tail fiber domains.Biophys Rev. 2018 Apr;10(2):463-471. doi: 10.1007/s12551-017-0348-5. Epub 2017 Dec 4. Biophys Rev. 2018. PMID: 29204885 Free PMC article. Review.
-
Morphogenesis of the T4 tail and tail fibers.Virol J. 2010 Dec 3;7:355. doi: 10.1186/1743-422X-7-355. Virol J. 2010. PMID: 21129200 Free PMC article. Review.
References
-
- Beckendorf, S. K. 1973. Structure of distal half of bacteriophage-T4 tail fiber. J. Mol. Biol. 73:37-53. - PubMed
-
- Bishop, R. J., and W. B. Wood. 1976. Genetic analysis of T4 tail fiber assembly. I. A gene 37 mutation that allows bypass of gene 38 function. Virology 72:244-254. - PubMed
-
- Boice, J. A., G. R. Dieckmann, W. F. DeGrado, and R. Fairman. 1996. Thermodynamic analysis of a designed three-stranded coiled coil. Biochemistry 35:14480-14485. - PubMed
-
- Cerritelli, M. E., J. S. Wall, M. N. Simon, J. F. Conway, and A. C. Steven. 1996. Stoichiometry and domainal organization of the long tail-fiber of bacteriophage T4: a hinged viral adhesin. J. Mol. Biol. 260:767-780. - PubMed
-
- Dickson, R. C. 1973. Assembly of bacteriophage T4 tail fibers. IV. J. Mol. Biol. 79:633-647. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

