Diversity and recognition efficiency of T cell responses to cancer
- PMID: 15578105
- PMCID: PMC529423
- DOI: 10.1371/journal.pmed.0010028
Diversity and recognition efficiency of T cell responses to cancer
Abstract
Background: Melanoma patients vaccinated with tumor-associated antigens frequently develop measurable peptide-specific CD8+ T cell responses; however, such responses often do not confer clinical benefit. Understanding why vaccine-elicited responses are beneficial in some patients but not in others will be important to improve targeted cancer immunotherapies.
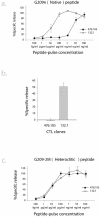
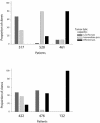
Methods and findings: We analyzed peptide-specific CD8+ T cell responses in detail, by generating and characterizing over 200 cytotoxic T lymphocyte clones derived from T cell responses to heteroclitic peptide vaccination, and compared these responses to endogenous anti-tumor T cell responses elicited naturally (a heteroclitic peptide is a modification of a native peptide sequence involving substitution of an amino acid at an anchor residue to enhance the immunogenicity of the peptide). We found that vaccine-elicited T cells are diverse in T cell receptor variable chain beta expression and exhibit a different recognition profile for heteroclitic versus native peptide. In particular, vaccine-elicited T cells respond to native peptide with predominantly low recognition efficiency--a measure of the sensitivity of a T cell to different cognate peptide concentrations for stimulation--and, as a result, are inefficient in tumor lysis. In contrast, endogenous tumor-associated-antigen-specific T cells show a predominantly high recognition efficiency for native peptide and efficiently lyse tumor targets.
Conclusions: These results suggest that factors that shape the peptide-specific T cell repertoire after vaccination may be different from those that affect the endogenous response. Furthermore, our findings suggest that current heteroclitic peptide vaccination protocols drive expansion of peptide-specific T cells with a diverse range of recognition efficiencies, a significant proportion of which are unable to respond to melanoma cells. Therefore, it is critical that the recognition efficiency of vaccine-elicited T cells be measured, with the goal of advancing those modalities that elicit T cells with the greatest potential of tumor reactivity.
Conflict of interest statement
Figures



Comment in
-
Tumor cell recognition efficiency by T cells.PLoS Med. 2005 Mar;2(3):e77; author reply e95. doi: 10.1371/journal.pmed.0020077. Epub 2005 Mar 29. PLoS Med. 2005. PMID: 15783261 Free PMC article. No abstract available.
References
-
- Yee C, Thompson JA, Byrd D, Riddell SR, Roche P, et al. Adoptive T cell therapy using antigen-specific CD8+ T cell clones for the treatment of patients with metastatic melanoma: In vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci U S A. 2002;99:16168–16173. - PMC - PubMed
-
- Lee PP, Yee C, Savage PA, Fong L, Brockstedt D, et al. Characterization of circulating T cells specific for tumor-associated antigens in melanoma patients. Nat Med. 1999;5:677–685. - PubMed
-
- Jonuleit H, Giesecke-Tuettenberg A, Tuting T, Thurner-Schuler B, Stuge TB, et al. A comparison of two types of dendritic cell as adjuvants for the induction of melanoma-specific T-cell responses in humans following intranodal injection. Int J Cancer. 2001;93:243–251. - PubMed
-
- Lee P, Wang F, Kuniyoshi J, Rubio V, Stuge T, et al. Effects of interleukin-12 on the immune response to a multipeptide vaccine for resected metastatic melanoma. J Clin Oncol. 2001;19:3836–3847. - PubMed
-
- Weber J, Sondak VK, Scotland R, Phillip R, Wang F, et al. Granulocyte-macrophage-colony-stimulating factor added to a multipeptide vaccine for resected stage II melanoma. Cancer. 2003;97:186–200. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

