Otopetrin 1 is required for otolith formation in the zebrafish Danio rerio
- PMID: 15581873
- PMCID: PMC2522322
- DOI: 10.1016/j.ydbio.2004.09.001
Otopetrin 1 is required for otolith formation in the zebrafish Danio rerio
Abstract
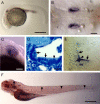
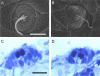
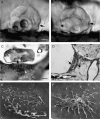
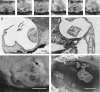
Orientation with respect to gravity is essential for the survival of complex organisms. The gravity receptor is one of the phylogenetically oldest sensory systems, and special adaptations that enhance sensitivity to gravity are highly conserved. The fish inner ear contains three large extracellular biomineral particles, otoliths, which have evolved to transduce the force of gravity into neuronal signals. Mammalian ears contain thousands of small particles called otoconia that serve a similar function. Loss or displacement of these structures can be lethal for fish and is responsible for benign paroxysmal positional vertigo (BPPV) in humans. The distinct morphologies of otoconial particles and otoliths suggest divergent developmental mechanisms. Mutations in a novel gene Otopetrin 1 (Otop1), encoding multi-transmembrane domain protein, result in nonsyndromic otoconial agenesis and a severe balance disorder in mice. Here we show that the zebrafish, Danio rerio, contains a highly conserved gene, otop1, that is essential for otolith formation. Morpholino-mediated knockdown of zebrafish Otop1 leads to otolith agenesis without affecting the sensory epithelium or other structures within the inner ear. Despite lack of otoliths in early development, otolith formation partially recovers in some fish after 2 days. However, the otoliths are malformed, misplaced, lack an organic matrix, and often consist of inorganic calcite crystals. These studies demonstrate that Otop1 has an essential and conserved role in the timing of formation and the size and shape of the developing otolith.
Figures



References
-
- Addadi L, Berman A, Oldak JM, Weiner S. Structural and stereochemical relations between acidic macromolecules of organic matrices and crystals. Connect. Tissue Res. 1989;21:127–134. - PubMed
-
- Bever MM, Fekete DM. Atlas of the developing inner ear in zebrafish. Dev. Dyn. 2002;223:536–543. - PubMed
-
- Borelli G, Mayer-Gostan N, Merle PL, Pontual H, Boeuf G, Allemand D, Payan P. Composition of biomineral organic matrices with special emphasis on turbot (Psetta maxima) otolith and endolymph. Calcif. Tissue Int. 2003;72:717–725. - PubMed
-
- Carlstrom D, Engstrom H, Hjorth S. Electron microscopic and X-ray diffraction studies of statoconia. Laryngoscope. 1953;63:1052–1057. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

