Structure of Ca2+ release channel at 14 A resolution
- PMID: 15581887
- PMCID: PMC2978512
- DOI: 10.1016/j.jmb.2004.10.073
Structure of Ca2+ release channel at 14 A resolution
Abstract
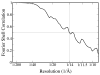
The 14 A resolution structure of the 2.3 MDa Ca2+ release channel (also known as RyR1) was determined by electron cryomicroscopy and single particle reconstruction. This structure was produced using collected data used for our previous published structures at 22-30 A resolution, but now taking advantage of recent algorithmic improvements in the EMAN software suite. This improved map clearly exhibits more structural detail and allows better defined docking of computationally predicted structural domain folds. Using sequence-based fold recognition, the N-terminal region of RyR1, residues 216-572, was predicted to have significant structural similarity with the IP3-binding core region of the type 1 IP3R. This putative structure was computationally localized to the clamp-shaped region of RyR1, which has been implicated to have a regulatory role in the channel activity.
Figures


References
-
- Serysheva II, Orlova EV, Chiu W, Sherman MB, Hamilton SL, Van Heel M. Electron cryomicroscopy and angular reconstitution used to visualize the skeletal muscle calcium release channel. Nature Struct Biol. 1995;2:18–24. - PubMed
-
- Orlova EV, Serysheva II, van Heel M, Hamilton SL, Chiu W. Two structural configurations of the skeletal muscle calcium release channel. Nature Struct Biol. 1996;3:547–552. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

