Ezrin-radixin-moesin (ERM)-binding phosphoprotein 50 organizes ERM proteins at the apical membrane of polarized epithelia
- PMID: 15591354
- PMCID: PMC539771
- DOI: 10.1073/pnas.0407974101
Ezrin-radixin-moesin (ERM)-binding phosphoprotein 50 organizes ERM proteins at the apical membrane of polarized epithelia
Abstract
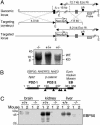
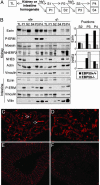
Ezrin-radixin-moesin (ERM) proteins regulate the organization and function of specific cortical structures in polarized epithelial cells by connecting filamentous (F)-actin to plasma membrane proteins. The contribution of ERM proteins to these structures depends on a conformational change to an active state in which the C-terminal region interacts with F-actin and the N-terminal domain interacts with membrane ligands. The specific ligands necessary for stabilizing ERM proteins at the membrane are not known. By generating mice deficient for ERM-binding phosphoprotein 50/Na(+)/H(+) exchanger regulatory factor 1 (EBP50/NHERF1), which binds the N-terminal domain of ERM proteins, we found that EBP50 is required for the maintenance of active ERM proteins at the cortical brush border membranes (BBM) of polarized epithelia. In EBP50(-/-) mice, ERM proteins were significantly decreased specifically in BBM from kidney and small intestine epithelial cells, whereas they remained unchanged in the cytoplasm. In wild-type animals, EBP50 was localized to the BBM compartment where it was processed by cleavage of the ERM-binding motif. In BBM, active ERM proteins formed distinct complexes with full-length EBP50 and with F-actin, suggesting a switch mechanism in which proteolytically processed EBP50 would release ERM proteins to complex with F-actin. The structural defects found in the EBP50(-/-) intestinal microvilli were reminiscent of those described in ezrin(-/-) mice, suggesting a role for EBP50 in organizing apical epithelial membranes.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

