A two-photon FRAP analysis of the cytoskeleton dynamics in the microvilli of intestinal cells
- PMID: 15596489
- PMCID: PMC1305148
- DOI: 10.1529/biophysj.104.049619
A two-photon FRAP analysis of the cytoskeleton dynamics in the microvilli of intestinal cells
Abstract
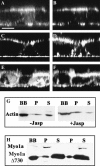
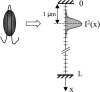
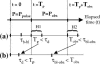
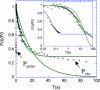
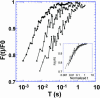
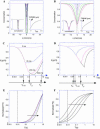
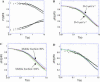
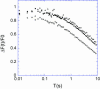
The molecular structure of the brush-border of enterocytes has been investigated since the 1980s, but the dynamics of this highly specialized subcellular domain have been difficult to study due to its small size. To perform a detailed analysis of the dynamics of cytoskeleton proteins in this domain, we developed two-photon fluorescence recovery after photobleaching and a theoretical framework for data analysis. With this method, fast dynamics of proteins in the microvilli of the brush border of epithelial intestinal cells can be measured on the millisecond timescale in volumes smaller than 1 microm3. Two major proteins of the cytoskeleton of the microvilli, actin and myosin 1a (Myo1a; formerly named brush border myosin I), are mobile in the brush-border of Caco-2 cells, an enterocyte-like cellular model. However, the mobility of actin is very different from that of Myo1a and they appear to be unrelated (diffusion coefficient of 15 microm2 s(-1) with a mobile fraction of 60% for actin, and 4 microm2 s(-1) with a mobile fraction of 90% for Myo1a). Furthermore, we show for the first time, in vivo, that the dynamics of Myo1a in microvilli reflect its motor activity.
Figures


Similar articles
-
Differential localization and dynamics of class I myosins in the enterocyte microvillus.Mol Biol Cell. 2010 Mar 15;21(6):970-8. doi: 10.1091/mbc.e09-07-0638. Epub 2010 Jan 20. Mol Biol Cell. 2010. PMID: 20089841 Free PMC article.
-
Myosin-1A targets to microvilli using multiple membrane binding motifs in the tail homology 1 (TH1) domain.J Biol Chem. 2012 Apr 13;287(16):13104-15. doi: 10.1074/jbc.M111.336313. Epub 2012 Feb 24. J Biol Chem. 2012. PMID: 22367206 Free PMC article.
-
Dynamics and morphology of microvilli driven by actin polymerization.Phys Rev Lett. 2006 Jul 7;97(1):018101. doi: 10.1103/PhysRevLett.97.018101. Epub 2006 Jul 5. Phys Rev Lett. 2006. PMID: 16907410
-
Shaping the intestinal brush border.J Cell Biol. 2014 Nov 24;207(4):441-51. doi: 10.1083/jcb.201407015. J Cell Biol. 2014. PMID: 25422372 Free PMC article. Review.
-
Vesicle transport: the role of actin filaments and myosin motors.Microsc Res Tech. 1999 Oct 15;47(2):93-106. doi: 10.1002/(SICI)1097-0029(19991015)47:2<93::AID-JEMT2>3.0.CO;2-P. Microsc Res Tech. 1999. PMID: 10523788 Review.
Cited by
-
Profilin-Mediated Actin Allocation Regulates the Growth of Epithelial Microvilli.Curr Biol. 2019 Oct 21;29(20):3457-3465.e3. doi: 10.1016/j.cub.2019.08.051. Epub 2019 Oct 10. Curr Biol. 2019. PMID: 31607529 Free PMC article.
-
Mechanosensitive Adaptation of E-Cadherin Turnover across adherens Junctions.PLoS One. 2015 Jun 5;10(6):e0128281. doi: 10.1371/journal.pone.0128281. eCollection 2015. PLoS One. 2015. PMID: 26046627 Free PMC article.
-
PDZ interactions regulate rapid turnover of the scaffolding protein EBP50 in microvilli.J Cell Biol. 2012 Jul 23;198(2):195-203. doi: 10.1083/jcb.201204008. Epub 2012 Jul 16. J Cell Biol. 2012. PMID: 22801783 Free PMC article.
-
Evidence for a common mode of transcription factor interaction with chromatin as revealed by improved quantitative fluorescence recovery after photobleaching.Biophys J. 2008 Apr 15;94(8):3323-39. doi: 10.1529/biophysj.107.123182. Epub 2008 Jan 16. Biophys J. 2008. PMID: 18199661 Free PMC article.
-
Intravital microscopy: a practical guide on imaging intracellular structures in live animals.Bioarchitecture. 2012 Sep-Oct;2(5):143-57. doi: 10.4161/bioa.21758. Epub 2012 Sep 1. Bioarchitecture. 2012. PMID: 22992750 Free PMC article. Review.
References
-
- Bacallao, R., A. Garfinkel, S. Monke, G. Zampighi, and L. J. Mandel. 1994. ATP depletion: a novel method to study junctional properties in epithelial tissues. I. Rearrangement of the actin cytoskeleton. J. Cell Sci. 107:3301–3313. - PubMed
-
- Ballestrem, C., B. Wehrle-Haller, and B. A. Imhof. 1998. Actin dynamics in living mammalian cells. J. Cell Sci. 111:1649–1658. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

