The cytoplasmic tail slows the folding of human immunodeficiency virus type 1 Env from a late prebundle configuration into the six-helix bundle
- PMID: 15596806
- PMCID: PMC538707
- DOI: 10.1128/JVI.79.1.106-115.2005
The cytoplasmic tail slows the folding of human immunodeficiency virus type 1 Env from a late prebundle configuration into the six-helix bundle
Abstract
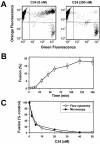
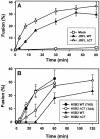
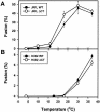
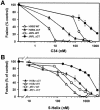
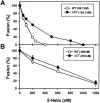
Effects of the cytoplasmic tail (CT) of human immunodeficiency virus type 1 Env on the process of membrane fusion were investigated. Full-length Env (wild type [WT]) and Env with its CT truncated (DeltaCT) were expressed on cell surfaces, these cells were fused to target cells, and the inhibition of fusion by peptides that prevent Env from folding into a six-helix bundle conformation was measured. For both X4-tropic and R5-tropic Env proteins, DeltaCT induced faster fusion kinetics than did the WT, and peptides were less effective at inhibiting DeltaCT-induced fusion. We tested the hypothesis that the inhibitory peptides were less effective at inhibiting DeltaCT-induced fusion because DeltaCT folds more quickly into a six-helix bundle. Early and late intermediates of WT- and DeltaCT-induced fusion were captured, and the ability of peptides to block fusion when added at the intermediate stages was quantified. When added at the early intermediate, the peptides were still less effective at inhibiting DeltaCT-induced fusion but they were equally effective at preventing WT- and DeltaCT-induced fusion when added at the late intermediate. We conclude that for both X4-tropic and R5-tropic Env proteins, the CT facilitates conformational changes that allow the trimeric coiled coil of prebundles to become optimally exposed. But once Env does favorably expose its coiled coil to inhibitory peptides, the CT hinders subsequent folding into a six-helix bundle. Because of this facilitation of maximal exposure and hindrance of bundle formation, the coiled coil is optimally exposed for a longer time for WT than for DeltaCT. This accounts for the greater peptide inhibition of WT-induced fusion.
Figures


References
-
- Armand-Ugon, M., A. Gutierrez, B. Clotet, and J. A. Este. 2003. HIV-1 resistance to the gp41-dependent fusion inhibitor C-34. Antiviral Res. 59:137-142. - PubMed
-
- Blacklow, S. C., M. Lu, and P. S. Kim. 1995. A trimeric subdomain of the simian immunodeficiency virus envelope glycoprotein. Biochemistry 34:14955-14962. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

