Rad23 stabilizes Rad4 from degradation by the Ub/proteasome pathway
- PMID: 15601997
- PMCID: PMC545455
- DOI: 10.1093/nar/gkh987
Rad23 stabilizes Rad4 from degradation by the Ub/proteasome pathway
Abstract
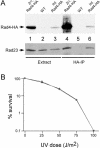
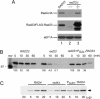
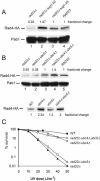
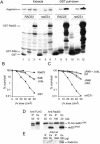
Rad23 protein interacts with the nucleotide excision-repair (NER) factor Rad4, and the dimer can bind damaged DNA. Rad23 also binds ubiquitinated proteins and promotes their degradation by the proteasome. Rad23/proteasome interaction is required for efficient NER, although the specific role of the Ub/proteasome system in DNA repair is unclear. We report that the availability of Rad4 contributes significantly to the cellular tolerance to UV light. Mutations in the proteasome, and in genes encoding the ubiquitin-conjugating enzymes Ubc4 and Ubc5, stabilized Rad4 and increased tolerance to UV light. A short amino acid sequence, previously identified in human Rad23, mediates the interaction between Rad23 and Rad4. We determined that this motif was required for stabilizing Rad4, and could function independently of the intact protein. A ubiquitin-like (UbL) domain in Rad23 binds the proteasome, and is required for conferring full resistance to DNA damage. However, Rad23/proteasome interaction appears unrelated to Rad23-mediated stabilization of Rad4. Specifically, simultaneous expression of a Rad23 mutant that could not bind the proteasome, with a mutant that could not interact with Rad4, fully suppressed the UV sensitivity of rad23Delta, demonstrating that Rad23 performs two independent, but concurrent roles in NER.
Figures


Similar articles
-
Distinct functions of the ubiquitin-proteasome pathway influence nucleotide excision repair.EMBO J. 2006 Jun 7;25(11):2529-38. doi: 10.1038/sj.emboj.7601120. EMBO J. 2006. PMID: 16675952 Free PMC article.
-
Yeast deubiquitinase Ubp3 interacts with the 26 S proteasome to facilitate Rad4 degradation.J Biol Chem. 2010 Nov 26;285(48):37542-50. doi: 10.1074/jbc.M110.170175. Epub 2010 Sep 27. J Biol Chem. 2010. PMID: 20876584 Free PMC article.
-
Roles of Rad23 protein in yeast nucleotide excision repair.Nucleic Acids Res. 2004 Nov 15;32(20):5981-90. doi: 10.1093/nar/gkh934. Print 2004. Nucleic Acids Res. 2004. PMID: 15545636 Free PMC article.
-
The ubiquitin receptor Rad23: at the crossroads of nucleotide excision repair and proteasomal degradation.DNA Repair (Amst). 2009 Apr 5;8(4):449-60. doi: 10.1016/j.dnarep.2009.01.005. Epub 2009 Feb 14. DNA Repair (Amst). 2009. PMID: 19223247 Review.
-
The moonlighting of RAD23 in DNA repair and protein degradation.Biochim Biophys Acta Gene Regul Mech. 2023 Jun;1866(2):194925. doi: 10.1016/j.bbagrm.2023.194925. Epub 2023 Feb 28. Biochim Biophys Acta Gene Regul Mech. 2023. PMID: 36863450 Review.
Cited by
-
Distinct functions of the ubiquitin-proteasome pathway influence nucleotide excision repair.EMBO J. 2006 Jun 7;25(11):2529-38. doi: 10.1038/sj.emboj.7601120. EMBO J. 2006. PMID: 16675952 Free PMC article.
-
Divergent roles of Rad4 and Rad23 homologs in Metarhizium robertsii's resistance to solar ultraviolet damage.Appl Environ Microbiol. 2023 Sep 28;89(9):e0099423. doi: 10.1128/aem.00994-23. Epub 2023 Sep 1. Appl Environ Microbiol. 2023. PMID: 37655890 Free PMC article.
-
UV induced ubiquitination of the yeast Rad4-Rad23 complex promotes survival by regulating cellular dNTP pools.Nucleic Acids Res. 2015 Sep 3;43(15):7360-70. doi: 10.1093/nar/gkv680. Epub 2015 Jul 6. Nucleic Acids Res. 2015. PMID: 26150418 Free PMC article.
-
Rad1 and Rad10 Tied to Photolyase Regulators Protect Insecticidal Fungal Cells from Solar UV Damage by Photoreactivation.J Fungi (Basel). 2022 Oct 25;8(11):1124. doi: 10.3390/jof8111124. J Fungi (Basel). 2022. PMID: 36354891 Free PMC article.
-
Rad23 interaction with the proteasome is regulated by phosphorylation of its ubiquitin-like (UbL) domain.J Mol Biol. 2014 Dec 12;426(24):4049-4060. doi: 10.1016/j.jmb.2014.10.004. Epub 2014 Oct 13. J Mol Biol. 2014. PMID: 25311859 Free PMC article.
References
-
- Batty D.P. and Wood,R.D. (2000) Damage recognition in nucleotide excision repair of DNA2. Gene, 241, 193–204. - PubMed
-
- Prakash S. and Prakash,L. (2000) Nucleotide excision repair in yeast. Mutat. Res., 451, 13–24. - PubMed
-
- Elsasser S., Chandler-Militello,D., Muller,B., Hanna,J. and Finley,D. (2004) Rad23 and Rpn10 serve as alternative ubiquitin receptors for the proteasome. J. Biol. Chem., 279, 26817–26822. - PubMed
-
- Schauber C., Chen,L., Tongaonkar,P., Vega,I., Lambertson,D., Potts,W. and Madura,K. (1998) Rad23 links DNA repair to the ubiquitin/proteasome pathway. Nature, 391, 715–718. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

