TBX21: a functional variant predicts improvement in asthma with the use of inhaled corticosteroids
- PMID: 15604153
- PMCID: PMC539815
- DOI: 10.1073/pnas.0408532102
TBX21: a functional variant predicts improvement in asthma with the use of inhaled corticosteroids
Abstract
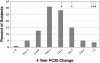
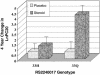
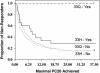
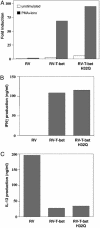
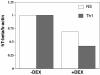
TBX21 encodes for the transcription factor T-bet (T-box expressed in T cells), which influences naive T lymphocyte development and has been implicated in asthma pathogenesis. Specifically, the T-bet knockout mouse spontaneously develops airway hyperresponsiveness and other changes consistent with asthma. Because airway responsiveness is moderated by the use of inhaled corticosteroids in asthma, it is conceivable that genetic variation in TBX21 may alter asthma phenotypes in a treatment-specific fashion. Here we demonstrate that the nonsynonymous variation in TBX21 coding for replacement of histidine 33 with glutamine is associated with significant improvement in the PC(20) (a measure of airway responsiveness) of asthmatic children in a large clinical trial spanning 4 years. We note that this increase occurs only in the children randomized to inhaled corticosteroids and that it dramatically enhances the overall improvement in PC(20) associated with inhaled corticosteroid usage. The average PC(20) at trial end for subjects on inhaled corticosteroids possessing a variant allele was in the normal range for nonasthmatics. In cellular models, we show that the TBX21 variant increases T helper 1 and decreases T helper 2 cytokine expression comparably with wild type. TBX21 may thus be an important determinant pharmacogenetic response to the therapy of asthma with inhaled corticosteroids.
Figures

Similar articles
-
Pharmacogenetic study of the effects of NK2R G231E G>A and TBX21 H33Q C>G polymorphisms on asthma control with inhaled corticosteroid treatment.J Clin Pharm Ther. 2009 Dec;34(6):693-701. doi: 10.1111/j.1365-2710.2009.01054.x. J Clin Pharm Ther. 2009. PMID: 20175803
-
Genetic variation of TBX21 gene increases risk of asthma and its severity in Indian children.J Hum Genet. 2014 Aug;59(8):437-43. doi: 10.1038/jhg.2014.52. Epub 2014 Jul 24. J Hum Genet. 2014. PMID: 25056814
-
Genome-wide association identifies the T gene as a novel asthma pharmacogenetic locus.Am J Respir Crit Care Med. 2012 Jun 15;185(12):1286-91. doi: 10.1164/rccm.201111-2061OC. Epub 2012 Apr 26. Am J Respir Crit Care Med. 2012. PMID: 22538805 Free PMC article. Clinical Trial.
-
Effects of genetic factors to inhaled corticosteroid response in children with asthma: a literature review.J Int Med Res. 2017 Dec;45(6):1818-1830. doi: 10.1177/0300060516683877. Epub 2017 Jan 25. J Int Med Res. 2017. PMID: 29251255 Free PMC article. Review.
-
Inhaled corticosteroid therapy in newly detected mild asthma.Drugs. 1999;58 Suppl 4:17-24; discussion 52. doi: 10.2165/00003495-199958004-00003. Drugs. 1999. PMID: 10711855 Review.
Cited by
-
Relationship between the benefits of suplatast tosilate, a Th2 cytokine inhibitor, and gene polymorphisms in children with bronchial asthma.Exp Ther Med. 2010 Nov;1(6):977-982. doi: 10.3892/etm.2010.149. Epub 2010 Sep 15. Exp Ther Med. 2010. PMID: 22993628 Free PMC article.
-
Pharmacogenetics and interstitial lung disease.Curr Opin Pulm Med. 2016 Sep;22(5):456-65. doi: 10.1097/MCP.0000000000000289. Curr Opin Pulm Med. 2016. PMID: 27253772 Free PMC article. Review.
-
Pharmacogenomic Response of Inhaled Corticosteroids for the Treatment of Asthma: Considerations for Therapy.Pharmgenomics Pers Med. 2020 Aug 4;13:261-271. doi: 10.2147/PGPM.S231471. eCollection 2020. Pharmgenomics Pers Med. 2020. PMID: 32801837 Free PMC article. Review.
-
The pharmacogenetics research network: from SNP discovery to clinical drug response.Clin Pharmacol Ther. 2007 Mar;81(3):328-45. doi: 10.1038/sj.clpt.6100087. Clin Pharmacol Ther. 2007. PMID: 17339863 Free PMC article. Review.
-
Pharmacogenomics of pediatric asthma.Indian J Hum Genet. 2010 Sep;16(3):111-8. doi: 10.4103/0971-6866.73398. Indian J Hum Genet. 2010. PMID: 21206697 Free PMC article.
References
-
- Barnes, P. J. (1996) Am. J. Respir. Crit. Care Med. 154, S21–S27. - PubMed
-
- Bousquet, J. (2000) Clin. Exp. Allergy 30, Suppl. 1, 2–5. - PubMed
-
- Lemanske, R. F., Jr., & Allen, D. B. (1997) Am. J. Respir. Crit. Care Med. 156, 685–687. - PubMed
-
- Malmstrom, K., Rodriguez-Gomez, G., Guerra, J., Villaran, C., Pineiro, A., Wei, L. X., Seidenberg, B. C. & Reiss, T. F. (1999) Ann. Intern. Med. 130, 487–495. - PubMed
-
- Szefler, S. J., Martin, R. J., King, T. S., Boushey, H. A., Cherniack, R. M., Chinchilli, V. M., Craig, T. J., Dolovich, M., Drazen, J. M., Fagan, J. K., et al. (2002) J. Allergy Clin. Immunol. 109, 410–418. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- N01 HR016047/HL/NHLBI NIH HHS/United States
- HR16046/HR/NHLBI NIH HHS/United States
- N01 HR16044/HR/NHLBI NIH HHS/United States
- U19 AI031541/AI/NIAID NIH HHS/United States
- HR16051/HR/NHLBI NIH HHS/United States
- N01 HR016046/HL/NHLBI NIH HHS/United States
- HR16052/HR/NHLBI NIH HHS/United States
- HR16045/HR/NHLBI NIH HHS/United States
- HR16050/HR/NHLBI NIH HHS/United States
- N01 HR016049/HL/NHLBI NIH HHS/United States
- N01 HR016052/HL/NHLBI NIH HHS/United States
- AI31541/AI/NIAID NIH HHS/United States
- N01-HR-16049/HR/NHLBI NIH HHS/United States
- U01 HL65899/HL/NHLBI NIH HHS/United States
- U01 HL065899/HL/NHLBI NIH HHS/United States
- N01 HR016044/HL/NHLBI NIH HHS/United States
- HR16048/HR/NHLBI NIH HHS/United States
- N01 HR016050/HL/NHLBI NIH HHS/United States
- HR16049/HR/NHLBI NIH HHS/United States
- N01 HR016051/HL/NHLBI NIH HHS/United States
- N01 HR016045/HL/NHLBI NIH HHS/United States
- P01 AI031541/AI/NIAID NIH HHS/United States
- N01 HR016048/HL/NHLBI NIH HHS/United States
- HR16047/HR/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

