The novel parainfluenza virus hemagglutinin-neuraminidase inhibitor BCX 2798 prevents lethal synergism between a paramyxovirus and Streptococcus pneumoniae
- PMID: 15616320
- PMCID: PMC538863
- DOI: 10.1128/AAC.49.1.398-405.2005
The novel parainfluenza virus hemagglutinin-neuraminidase inhibitor BCX 2798 prevents lethal synergism between a paramyxovirus and Streptococcus pneumoniae
Abstract
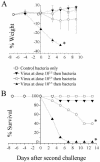
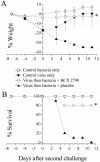
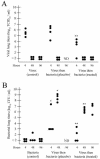
An association exists between respiratory viruses and bacterial infections. Prevention or treatment of the preceding viral infection is a logical goal for reducing this important cause of morbidity and mortality. The ability of the novel, selective parainfluenza virus hemagglutinin-neuraminidase inhibitor BCX 2798 to prevent the synergism between a paramyxovirus and Streptococcus pneumoniae was examined in this study. A model of secondary bacterial pneumonia after infection with a recombinant Sendai virus whose hemagglutinin-neuraminidase gene was replaced with that of human parainfluenza virus type 1 [rSV(hHN)] was established in mice. Challenge of mice with a sublethal dose of S. pneumoniae 7 days after a sublethal infection with rSV(hHN) (synergistic group) caused 100% mortality. Bacterial infection preceding viral infection had no effect on survival. The mean bacterial titers in the synergistic group were significantly higher than in mice infected with bacteria only. The virus titers were similar in mice infected with rSV(hHN) alone and in dually infected mice. Intranasal administration of BCX 2798 at 10 mg/kg per day to the synergistic group of mice starting 4 h before virus infection protected 80% of animals from death. This effect was accompanied by a significant reduction in lung viral and bacterial titers. Treatment of mice 24 h after the rSV(hHN) infection showed no protection against synergistic lethality. Together, our results indicate that parainfluenza viruses can prime for secondary bacterial infections. Prophylaxis of parainfluenza virus infections with antivirals might be an effective strategy for prevention of secondary bacterial complications in humans.
Figures


References
-
- Alymova, I. V., G. Taylor, T. Takimoto, T. H. Lin, P. Chand, Y. S. Babu, C. Li, X. Xiong, and A. Portner. 2004. Efficacy of novel hemagglutinin-neuraminidase inhibitors BCX 2798 and BCX 2855 against human parainfluenza viruses in vitro and in vivo. Antimicrob. Agents Chemother. 48:1495-1502. - PMC - PubMed
-
- Belshe, R. B., P. M. Mendelman, J. Treanor, J. King, W. C. Gruber, P. Piedra, D. I. Bernstein, F. G. Hayden, K. Kotloff, K. Zangwill, D. Iacuzio, and M. Wolff. 1998. The efficacy of live attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine in children. N. Engl. J. Med. 338:1405-1412. - PubMed
-
- Bousse, T., T. Takimoto, T. Matrosovich, and A. Portner. 2001. Two regions of the P protein are required to be active with the L protein for human parainfluenza virus type 1 RNA polymerase activity. Virology 283:306-314. - PubMed
-
- Chonmaitree, T., V. M. Howie, and A. L. Truant. 1986. Presence of respiratory viruses in middle ear fluids and nasal wash specimens from children with acute otitis media. Pediatrics 77:698-702. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

