Synthesis and conformational analysis of 6-C-methyl-substituted 2-acetamido-2-deoxy-beta-D-glucopyranosyl mono- and disaccharides
- PMID: 15624925
- PMCID: PMC1851687
- DOI: 10.1021/jo0485841
Synthesis and conformational analysis of 6-C-methyl-substituted 2-acetamido-2-deoxy-beta-D-glucopyranosyl mono- and disaccharides
Abstract
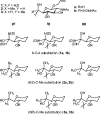
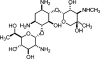
Several 6-C-substituted 2-acetamido-2-deoxy-beta-D-glucopyranosides (beta-D-GlcNAc monosaccharides 1a-3a and 1,4-linked disaccharides 1b-3b) were studied by solution NMR spectroscopy. Conformational analysis of the (6S)- and (6R)-C-methyl-substituted beta-d-GlcNAc monosaccharides indicates that the stereodefined methyl groups impose predictable conformational biases on the exocyclic C-5-C-6 bond, as determined by (1)H-(1)H and (13)C-(1)H coupling constants. Variable-temperature NMR experiments in methanol-d(4) were performed to determine DeltaDeltaH and DeltaDeltaS values derived from the two lowest energy conformers. These indicate that while the influence of 6-C-methyl substitution on conformational enthalpy is in accord with the classic principles of steric interactions, conformational preference in solution can also be strongly affected by other factors such as solvent-solute interactions and solvent reorganization.
Figures





Similar articles
-
Evaluation of steric effects on the exocyclic conformations of 6-C-methyl-substituted 2-acetamido-2-deoxy-beta-D-glucopyranosides.Carbohydr Res. 2002 Feb 5;337(2):83-6. doi: 10.1016/s0008-6215(01)00295-6. Carbohydr Res. 2002. PMID: 11814438
-
Conformational analysis of beta-glycosidic linkages in 13C-labeled glucobiosides using inter-residue scalar coupling constants.J Phys Chem B. 2008 Apr 10;112(14):4447-53. doi: 10.1021/jp710977k. Epub 2008 Mar 18. J Phys Chem B. 2008. PMID: 18345660
-
Synthesis of three hetero disaccharides, 4-O-beta-glucopyranosyl-6-deoxy-D-glucose, 4-O-beta-D-glucopyranosyl-D-mannosamine, and 4-O-beta-D-glucopyranosyl-D-mannose, and confirmation of their structures by C-13 NMR and MS.Biochem Biophys Res Commun. 1995 Sep 14;214(2):568-75. doi: 10.1006/bbrc.1995.2323. Biochem Biophys Res Commun. 1995. PMID: 7677766
-
Hydroxymethyl rotamer populations in disaccharides.J Org Chem. 2003 Jun 13;68(12):4615-30. doi: 10.1021/jo026913o. J Org Chem. 2003. PMID: 12790564
-
Synthesis and conformational analysis of novel N(OCH3)-linked disaccharide analogues.Chemistry. 2004 Mar 19;10(6):1433-44. doi: 10.1002/chem.200305587. Chemistry. 2004. PMID: 15034887
Cited by
-
Stereocontrolled synthesis of the D- and L-glycero-beta-D-manno-heptopyranosides and their 6-deoxy analogues. Synthesis of methyl alpha-l-rhamno-pyranosyl-(1-->3)-D-glycero-beta-D-manno-heptopyranosyl- (1-->3)-6-deoxy-glycero-beta-D-manno-heptopyranosyl-(1-->4)-alpha-L- rhamno-pyranoside, a tetrasaccharide subunit of the lipopolysaccharide from Plesimonas shigelloides.J Am Chem Soc. 2006 Jun 21;128(24):8078-86. doi: 10.1021/ja061594u. J Am Chem Soc. 2006. PMID: 16771524 Free PMC article.
References
-
- Hecht SM, editor. Bioorganic Chemistry: Carbohydrates. Oxford University Press; Oxford, UK: 1999.
-
- Adelhorst K, Bock K. Acta Chem. Scand. 1992;46:1114–1121. - PubMed
-
- Bock K, Duus JØ. J. Carbohydr. Chem. 1994;13:513–543.
-
- Blackwell J. In: Methods in Enzymology. Wood WA, Kellogg ST, editors. Vol. 161. Academic Press; San Diego, CA: 1988. pp. 435–442.
-
- Minke R, Blackwell J. J. Mol. Biol. 1978;120:167–181. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

