Nitromusk and polycyclic musk compounds as long-term inhibitors of cellular xenobiotic defense systems mediated by multidrug transporters
- PMID: 15626642
- PMCID: PMC1253704
- DOI: 10.1289/ehp.7301
Nitromusk and polycyclic musk compounds as long-term inhibitors of cellular xenobiotic defense systems mediated by multidrug transporters
Abstract
Synthetic musk compounds, widely used as fragrances in consumer products, have been detected in human tissue and, surprisingly, in aquatic organisms such as fish and mollusks. Although their persistence and potential to bioaccumulate are of concern, the toxicity and environmental risks of these chemicals are generally regarded as low. Here, however, we show that nitromusks and polycyclic musks inhibit the activity of multidrug efflux transporters responsible for multixenobiotic resistance (MXR) in gills of the marine mussel Mytilus californianus. The IC(subscript)10(/subscript) (concentration that inhibits 10%) values for the different classes of musks were in the range of 0.09-0.39 microM, and IC(subscript)50(/subscript) values were 0.74-2.56 microM. The immediate consequence of inhibition of efflux transporters is that normally excluded xenobiotics will now be able to enter the cell. Remarkably, the inhibitory effects of a brief 2-hr exposure to musks were only partially reversed after a 24- to 48-hr recovery period in clean seawater. This unexpected consequence of synthetic musks--a long-term loss of efflux transport activity--will result in continued accumulation of normally excluded toxicants even after direct exposure to the musk has ended. These findings also point to the need to determine whether other environmental chemicals have similar long-term effects on these transporters. The results are relevant to human health because they raise the possibility that exposure to common xenobiotics and pharmaceuticals could cause similar long-term inhibition of these transporters and lead to increased exposure to normally excluded toxicants.
Figures

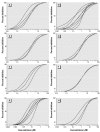
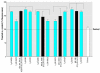
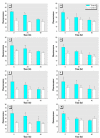

Comment in
-
Synthetic musks-arising new environmental menace?Mar Pollut Bull. 2005 Oct;50(10):1025-6. doi: 10.1016/j.marpolbul.2005.07.005. Mar Pollut Bull. 2005. PMID: 16112142 No abstract available.
-
Synthetic musk compounds and effects on human health?Environ Health Perspect. 2005 Dec;113(12):A802-3; author reply A803-4. doi: 10.1289/ehp.113-a802b. Environ Health Perspect. 2005. PMID: 16330332 Free PMC article. No abstract available.
References
-
- Api AM, Smith RL, Pipino S, Marczylo T, De Matteis F. Evaluation of the oral subchronic toxicity of AHTN (7-acetyl-1,1,3,4,4,6-hexamethyl-1,2,3,4-tetrahydronaphthalene) in the rat. Food Chem Toxicol. 2004;42(5):791–801. - PubMed
-
- Bain LJ, LeBlanc GA. Interaction of structurally diverse pesticides with the human MDR1 gene product P-glycoprotein. Toxicol Appl Pharmacol. 1996;141(1):288–298. - PubMed
-
- Balk F, Blok H, Salvito D. 2001. Environmental risks of musk fragrance ingredients. In: Pharmaceuticals and Personal Care Products in the Environment—Scientific and Regulatory Issues (Daughton CG, Jones-Lepp TL, eds). Washington, DC:American Chemical Society, 168–190.
-
- Balk F, Ford RA. Environmental risk assessment for the polycyclic musks AHTN and HHCB in the EU. I. Fate and exposure assessment. Toxicol Lett. 1999a;111(1–2):57–79. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

