Ro/SSA autoantibodies directly bind cardiomyocytes, disturb calcium homeostasis, and mediate congenital heart block
- PMID: 15630133
- PMCID: PMC2212767
- DOI: 10.1084/jem.20041859
Ro/SSA autoantibodies directly bind cardiomyocytes, disturb calcium homeostasis, and mediate congenital heart block
Abstract
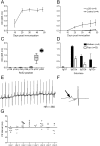
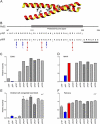
Congenital heart block develops in fetuses after placental transfer of Ro/SSA autoantibodies from rheumatic mothers. The condition is often fatal and the majority of live-born children require a pacemaker at an early age. The specific antibody that induces the heart block and the mechanism by which it mediates the pathogenic effect have not been elucidated. In this study, we define the cellular mechanism leading to the disease and show that maternal autoantibodies directed to a specific epitope within the leucine zipper amino acid sequence 200-239 (p200) of the Ro52 protein correlate with prolongation of fetal atrioventricular (AV) time and heart block. This finding was further confirmed experimentally in that pups born to rats immunized with p200 peptide developed AV block. p200-specific autoantibodies cloned from patients bound cultured cardiomyocytes and severely affected Ca2+ oscillations, leading to accumulating levels and overload of intracellular Ca2+ levels with subsequent loss of contractility and ultimately apoptosis. These findings suggest that passive transfer of maternal p200 autoantibodies causes congenital heart block by dysregulating Ca2+ homeostasis and inducing death in affected cells.
Figures


References
-
- Buyon, J.P., and R.M. Clancy. 2003. Neonatal lupus syndromes. Curr. Opin. Rheumatol. 15:535–541. - PubMed
-
- Brucato, A., M. Frassi, F. Franceschini, R. Cimaz, D. Faden, M.P. Pisoni, M. Muscara, G. Vignati, M. Stramba-Badiale, L. Catelli, et al. 2001. Risk of congenital complete heart block in newborns of mothers with anti-Ro/SSA antibodies detected by counterimmunoelectrophoresis: a prospective study of 100 women. Arthritis Rheum. 44:1832–1835. - PubMed
-
- Sonesson, S.E., S. Salomonsson, L.A. Jacobsson, K. Bremme, and M. Wahren-Herlenius. 2004. Signs of first-degree heart block occur in one-third of fetuses of pregnant women with anti-SSA/Ro 52-kd antibodies. Arthritis Rheum. 50:1253–1261. - PubMed
-
- Clancy, R.M., R.P. Kapur, Y. Molad, A.D. Askanase, and J.P. Buyon. 2004. Immunohistologic evidence supports apoptosis, IgG deposition, and novel macrophage/fibroblast crosstalk in the pathologic cascade leading to congenital heart block. Arthritis Rheum. 50:173–182. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

