Up-regulation of telomere-binding proteins, TRF1, TRF2, and TIN2 is related to telomere shortening during human multistep hepatocarcinogenesis
- PMID: 15632001
- PMCID: PMC1602303
- DOI: 10.1016/S0002-9440(10)62233-X
Up-regulation of telomere-binding proteins, TRF1, TRF2, and TIN2 is related to telomere shortening during human multistep hepatocarcinogenesis
Abstract
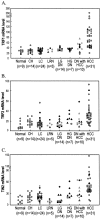
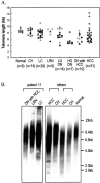
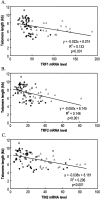
The telomeric repeat-binding factor 1 (TRF1), TRF2, and the TRF1-interacting nuclear protein 2 (TIN2) are involved in telomere maintenance. We describe the regulation of expression of these genes along with their relationship to telomere length in hepatocarcinogenesis. The transcriptional expression of these genes, TRF1 protein, and telomere length was examined in 9 normal livers, 14 chronic hepatitis, 24 liver cirrhosis, 5 large regenerative nodules, 14 low-grade dysplastic nodules (DNs), 7 high-grade DNs, 10 DNs with hepatocellular carcinoma (HCC) foci, and 31 HCCs. The expression of TRF1, TRF2, TIN2 mRNA, and TRF1 protein was gradually increased according to the progression of hepatocarcinogenesis with a marked increase in high-grade DNs and DNs with HCC foci and a further increase in HCCs. There was a gradual shortening of telomere during hepatocarcinogenesis with a significant reduction in length in DNs. Most nodular lesions (52 of 67) had shorter telomeres than their adjacent chronic hepatitis or liver cirrhosis, and the telomere lengths were inversely correlated with the mRNA level of these genes (P </= 0.001). This was more evident in DNs and DNs with HCC foci. In conclusion, TRF1, TRF2, and TIN2 might be involved in multistep hepatocarcinogenesis by playing crucial roles in telomere shortening.
Figures


Similar articles
-
Expression of telomeric repeat binding factor 1 and 2 and TRF1-interacting nuclear protein 2 in human gastric carcinomas.Int J Oncol. 2001 Sep;19(3):507-12. Int J Oncol. 2001. PMID: 11494028
-
Down-regulation of TRF1, TRF2 and TIN2 genes is important to maintain telomeric DNA for gastric cancers.Anticancer Res. 2002 Nov-Dec;22(6A):3303-7. Anticancer Res. 2002. PMID: 12530079
-
Coordinate regulation between expression levels of telomere-binding proteins and telomere length in breast carcinomas.Cancer Med. 2012 Oct;1(2):165-75. doi: 10.1002/cam4.14. Epub 2012 Jul 24. Cancer Med. 2012. PMID: 23342266 Free PMC article.
-
Early hepatocellular carcinoma and dysplastic nodules.Semin Liver Dis. 2005;25(2):133-42. doi: 10.1055/s-2005-871193. Semin Liver Dis. 2005. PMID: 15918142 Review.
-
Post-translational modifications of TRF1 and TRF2 and their roles in telomere maintenance.Mech Ageing Dev. 2012 Jun;133(6):421-34. doi: 10.1016/j.mad.2012.05.002. Epub 2012 May 23. Mech Ageing Dev. 2012. PMID: 22634377 Review.
Cited by
-
Telomere length modulation in human astroglial brain tumors.PLoS One. 2013 May 14;8(5):e64296. doi: 10.1371/journal.pone.0064296. Print 2013. PLoS One. 2013. PMID: 23691191 Free PMC article.
-
Cerium Oxide Nanoparticles Protect against Oxidant Injury and Interfere with Oxidative Mediated Kinase Signaling in Human-Derived Hepatocytes.Int J Mol Sci. 2019 Nov 27;20(23):5959. doi: 10.3390/ijms20235959. Int J Mol Sci. 2019. PMID: 31783479 Free PMC article.
-
Telomere Biology and Human Phenotype.Cells. 2019 Jan 19;8(1):73. doi: 10.3390/cells8010073. Cells. 2019. PMID: 30669451 Free PMC article. Review.
-
Cause-specific telomere factors deregulation in hepatocellular carcinoma.J Exp Clin Cancer Res. 2013 Sep 11;32(1):64. doi: 10.1186/1756-9966-32-64. J Exp Clin Cancer Res. 2013. PMID: 24020493 Free PMC article.
-
Telomere biology: Rationale for diagnostics and therapeutics in cancer.RNA Biol. 2015;12(10):1078-82. doi: 10.1080/15476286.2015.1081329. Epub 2015 Aug 20. RNA Biol. 2015. PMID: 26291128 Free PMC article. Review.
References
-
- Wright JH, Gottschling DE, Zakian VA. Saccharomyces telomeres assume a non-nucleosomal chromatin structure. Genes Dev. 1992;6:197–210. - PubMed
-
- Nugent CI, Hughes TR, Lue NF, Lundblad V. Cdc13p: a single-strand telomeric DNA-binding protein with a dual role in yeast telomere maintenance. Science. 1996;274:249–252. - PubMed
-
- Karlseder J. Telomere repeat binding factors: keeping the ends in check. Cancer Lett. 2003;194:189–197. - PubMed
-
- Collins K. Mammalian telomeres and telomerase. Curr Opin Cell Biol. 2000;12:378–383. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

