The MAP2/Tau family of microtubule-associated proteins
- PMID: 15642108
- PMCID: PMC549057
- DOI: 10.1186/gb-2004-6-1-204
The MAP2/Tau family of microtubule-associated proteins
Abstract
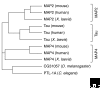
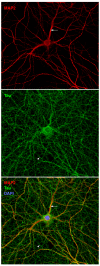
Microtubule-associated proteins (MAPs) of the MAP2/Tau family include the vertebrate proteins MAP2, MAP4, and Tau and homologs in other animals. All three vertebrate members of the family have alternative splice forms; all isoforms share a conserved carboxy-terminal domain containing microtubule-binding repeats, and an amino-terminal projection domain of varying size. MAP2 and Tau are found in neurons, whereas MAP4 is present in many other tissues but is generally absent from neurons. Members of the family are best known for their microtubule-stabilizing activity and for proposed roles regulating microtubule networks in the axons and dendrites of neurons. Contrary to this simple, traditional view, accumulating evidence suggests a much broader range of functions, such as binding to filamentous (F) actin, recruitment of signaling proteins, and regulation of microtubule-mediated transport. Tau is also implicated in Alzheimer's disease and other dementias. The ability of MAP2 to interact with both microtubules and F-actin might be critical for neuromorphogenic processes, such as neurite initiation, during which networks of microtubules and F-actin are reorganized in a coordinated manner. Various upstream kinases and interacting proteins have been identified that regulate the microtubule-stabilizing activity of MAP2/Tau family proteins.
Figures

References
-
- Hale CA, de Boer PA. Direct binding of FtsZ to ZipA, an essential component of the septal ring structure that mediates cell division in E. coli. Cell. 1997;88:175–185. doi: 10.1016/S0092-8674(00)81838-3. The interaction between the bacterial tubulin homolog FtsZ and an ancestral MAP, ZipA, is described. - DOI - PubMed
-
- RayChaudhuri D. ZipA is a MAP-Tau homolog and is essential for structural integrity of the cytokinetic FtsZ ring during bacterial cell division. EMBO J. 1999;18:2372–2383. doi: 10.1093/emboj/18.9.2372. The functional significance of the ancestral MAP ZipA in bacterial cell division is described and its relation to MAP2/Tau is proposed. - DOI - PMC - PubMed
-
- Hale CA, Rhee AC, de Boer PA. ZipA-induced bundling of FtsZ polymers mediated by an interaction between C-terminal domains. J Bacteriol. 2000;182:5153–5166. doi: 10.1128/JB.182.18.5153-5166.2000. The FtsZ interaction domain on ZipA is mapped to its carboxyl terminus, a region unrelated to MAP2/Tau, suggesting that ZipA is not a functional homolog of MAP2/Tau proteins. - DOI - PMC - PubMed
-
- Goedert M, Baur CP, Ahringer J, Jakes R, Hasegawa M, Spillantini MG, Smith MJ, Hill F. PTL-1, a microtubule-associated protein with tau-like repeats from the nematode Caenorhabditis elegans. J Cell Sci. 1996;109:2661–2672. Describes the cloning of a MAP2/Tau homolog from C. elegans, expression analyses, microtubule binding and stabilization experiments, and overexpression studies. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

