Postsynaptic shank antagonizes dendrite branching induced by the leucine-rich repeat protein Densin-180
- PMID: 15647492
- PMCID: PMC6725485
- DOI: 10.1523/JNEUROSCI.2699-04.2005
Postsynaptic shank antagonizes dendrite branching induced by the leucine-rich repeat protein Densin-180
Abstract
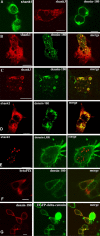
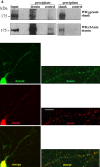
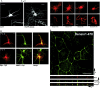
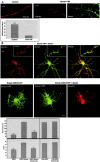
Leucine-rich repeat and PDZ [postsynaptic density-95 (PSD-95)/Discs large/zona occludens-1] domain proteins such as scribble and Densin-180 have been implicated in the establishment of cell-cell contacts. Here, we show that Densin-180, which has been identified as a constituent of the postsynaptic density in excitatory synapses interacts with the postsynaptic scaffold protein shank (shank1-3). The interaction involves a two-point attachment of the C-terminal region of Densin-180 with the Src homology 3 domain and the N-terminal part of the proline-rich region of shank proteins. The N-terminal leucine-rich repeat region, which is not involved in binding shank, targets Densin-180 to the plasma membrane in transfected cells and to the basolateral membrane of epithelial cells. Nevertheless, coexpression of shank leads to a redirection of Densin-180 into intracellular clusters. In cultured hippocampal neurons, Densin-180 overexpression induces excessive branching of neuronal dendrites, which occurs at the expense of clusters for the postsynaptic marker PSD-95. Coexpression of shank3 abrogates branch formation and targets Densin-180 into postsynaptic clusters instead. Shank blocks binding of delta-catenin but not alphaCaM kinase II to Densin-180; because delta-catenin has been shown to induce branching and neurite formation, our data suggest a mechanism where shank could block the activation of a Densin-180-dependent signaling pathway by delta-catenin.
Figures



Similar articles
-
Densin-180: revised membrane topology, domain structure and phosphorylation status.J Neurochem. 2009 Apr;109(2):297-302. doi: 10.1111/j.1471-4159.2009.05951.x. Epub 2009 Feb 2. J Neurochem. 2009. PMID: 19187442 Free PMC article. Review.
-
Densin-180 interacts with delta-catenin/neural plakophilin-related armadillo repeat protein at synapses.J Biol Chem. 2002 Feb 15;277(7):5345-50. doi: 10.1074/jbc.M110052200. Epub 2001 Nov 29. J Biol Chem. 2002. PMID: 11729199
-
A functional role of postsynaptic density-95-guanylate kinase-associated protein complex in regulating Shank assembly and stability to synapses.J Neurosci. 2004 Oct 20;24(42):9391-404. doi: 10.1523/JNEUROSCI.3314-04.2004. J Neurosci. 2004. PMID: 15496675 Free PMC article.
-
Densin-180, a synaptic protein, links to PSD-95 through its direct interaction with MAGUIN-1.Genes Cells. 2002 Nov;7(11):1149-60. doi: 10.1046/j.1365-2443.2002.00589.x. Genes Cells. 2002. PMID: 12390249
-
[Scaffold proteins (MAGUK, Shank and Homer) in postsynaptic density in the central nervous system].Postepy Biochem. 2007;53(2):188-97. Postepy Biochem. 2007. PMID: 17969881 Review. Polish.
Cited by
-
Rapid desensitization of the rat α7 nAChR is facilitated by the presence of a proline residue in the outer β-sheet.J Physiol. 2010 Nov 15;588(Pt 22):4415-29. doi: 10.1113/jphysiol.2010.195495. Epub 2010 Sep 13. J Physiol. 2010. PMID: 20837638 Free PMC article.
-
Densin-180: revised membrane topology, domain structure and phosphorylation status.J Neurochem. 2009 Apr;109(2):297-302. doi: 10.1111/j.1471-4159.2009.05951.x. Epub 2009 Feb 2. J Neurochem. 2009. PMID: 19187442 Free PMC article. Review.
-
The Neomycin Resistance Cassette in the Targeted Allele of Shank3B Knock-Out Mice Has Potential Off-Target Effects to Produce an Unusual Shank3 Isoform.Front Mol Neurosci. 2021 Jan 11;13:614435. doi: 10.3389/fnmol.2020.614435. eCollection 2020. Front Mol Neurosci. 2021. PMID: 33505245 Free PMC article.
-
Cell-intrinsic drivers of dendrite morphogenesis.Development. 2013 Dec;140(23):4657-71. doi: 10.1242/dev.087676. Development. 2013. PMID: 24255095 Free PMC article. Review.
-
Targeting of δ-catenin to postsynaptic sites through interaction with the Shank3 N-terminus.Mol Autism. 2020 Oct 28;11(1):85. doi: 10.1186/s13229-020-00385-8. Mol Autism. 2020. PMID: 33115499 Free PMC article.
References
-
- Bilder D, Perrimon N (2000) Localization of apical epithelial determinants by the basolateral PDZ protein Scribble. Nature 403: 676-680. - PubMed
-
- Bilder D, Schober M, Perrimon N (2003) Integrated activity of PDZ protein complexes regulates epithelial polarity. Nat Cell Biol 5: 53-58. - PubMed
-
- Böckers TM, Kreutz MR, Winter C, Zuschratter W, Smalla KH, Sanmarti-Vila L, Wex H, Langnaese K, Bockmann J, Garner CC, Gundelfinger ED (1999) Proline-rich synapse-associated protein-1/cortactin binding protein 1 (ProSAP1/CortBP1) is a PDZ-domain protein highly enriched in the postsynaptic density. J Neurosci 19: 6506-6518. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
